Visual Overview
Abstract
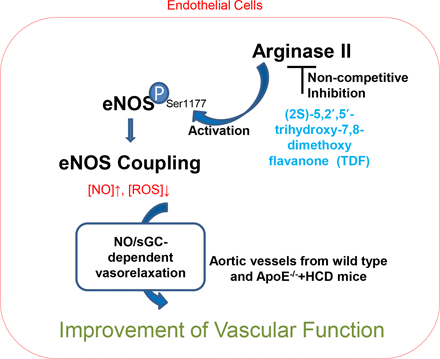
Elevated endothelial arginase activity decreases nitric oxide (NO) production by competing with the substrate l-arginine, previously reported, and reciprocally regulating endothelial nitric oxide synthase (eNOS) activity. Thus, arginase inhibitors may help treat vascular diseases associated with endothelial dysfunction. A screening of metabolites from medicinal plants revealed that (2S)-5,2′,5′-trihydroxy-7,8-dimethoxy flavanone (TDF) was a noncompetitive inhibitor of arginase. We investigated whether TDF reciprocally regulated endothelial NO production and its possible mechanism. TDF noncompetitively inhibited arginase I and II activity in a dose-dependent manner. TDF incubation decreased arginase activity and increased NO production in human umbilical vein endothelial cells and isolated mouse aortic vessels and reduced reactive oxygen species (ROS) generation in the endothelium of the latter. These TDF-mediated effects were associated with increased eNOS phosphorylation and dimerization but not with changes in protein content. Endothelium-dependent vasorelaxant responses to acetylcholine (Ach) were significantly increased in TDF-incubated aortic rings and attenuated by incubation with soluble guanylyl cyclase inhibitor. Phenylephrine-induced vasoconstrictor responses were markedly attenuated in TDF-treated vessels from wild-type mice. In atherogenic-prone ApoE−/− mice, TDF attenuated the high-cholesterol diet (HCD)-induced increase in arginase activity, which was accompanied by restoration of NO production and reduction of ROS generation. TDF incubation induced eNOS dimerization and phosphorylation at Ser1177. In addition, TDF improved Ach-dependent vasorelaxation responses and attenuated U46619-dependent contractile responses but did not change sodium nitroprusside–induced vasorelaxation or N-NAME-induced vasoconstriction. The findings suggest that TDF may help treat cardiovascular diseases by reducing pathophysiology derived from HCD-mediated endothelial dysfunction.
Footnotes
- Received March 23, 2015.
- Accepted August 7, 2015.
This work was supported by the Basic Science Research Program of the National Research Foundation of Korea, funded by the Ministry of Education, Science and Technology [Grant no. 2015R1A01017911] and [Grant no. 2014R1A6A1029617].
↵
 This article has supplemental material available at jpet.aspetjournals.org.
This article has supplemental material available at jpet.aspetjournals.org.
- Copyright © 2015 by The American Society for Pharmacology and Experimental Therapeutics
JPET articles become freely available 12 months after publication, and remain freely available for 5 years.Non-open access articles that fall outside this five year window are available only to institutional subscribers and current ASPET members, or through the article purchase feature at the bottom of the page.
|