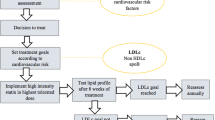
Abstract
A new class of compounds, known as squalene synthase inhibitors, has recently reached phase III clinical trials and may provide another therapeutic option for clinicians to improve risk management of low-density lipoprotein cholesterol (LDL-C). The clinical need for another LDL-C-lowering therapy is evident by the inability to achieve an LDL-C target of less than 70 mg/dL in the majority of very high-risk patients on statin monotherapy. Human clinical trial data with TAK-475, a novel and potent inhibitor of squalene synthase, have not yet been published.
Similar content being viewed by others
References and Recommended Reading
Grundy SM, Cleeman JI, Merz CN, et al.; National Heart, Lung, and Blood Institute; American College of Cardiology Foundation; American Heart Association: Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004, 110:227–239.
Smith SC Jr, Allen J, Blair SN, et al.; AHA/ACC; National Heart, Lung, and Blood Institute: AHA/ACC guidelines for secondary prevention for patients with coronary and other atherosclerotic vascular disease: 2006 update: endorsed by the National Heart, Lung, and Blood Institute. Circulation 2006, 113:2363–2372.
LaRosa JC, Grundy SM, Waters DD, et al.; Treating to New Targets (TNT) Investigators: Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med 2005, 352:1425–1435.
Pedersen TR, Faergeman O, Kastelein JJ, et al.; Incremental Decrease in End Points Through Aggressive Lipid Lowering (IDEAL) Study Group: High-dose atorvastatin vs usual-dose simvastatin for secondary prevention after myocardial infarction: the IDEAL study: a randomized controlled trial. JAMA 2005, 294:2437–2445.
Davidson MH, Maki KC, Pearson TA, et al.: Results of the National Cholesterol Education (NCEP) Program Evaluation ProjecT Utilizing Novel E-Technology (NEPTUNE) II survey and implications for treatment under the recent NCEP Writing Group recommendations. Am J Cardiol 2005, 96:556–563.
Nissen SE, Nicholls SJ, Sipahi I, et al.: ASTEROID Investigators: Effect of very high-intensity statin therapy on regression of coronary atherosclerosis: the ASTEROID trial. JAMA 2006, 295:1556–1565.
Robinson JG, Davidson MH: Combination therapy with ezetimibe and simvastatin to achieve aggressive LDL reduction. Expert Rev Cardiovasc Ther 2006, 4:461–476.
Davidson MH, Robinson JG: Lipid-lowering effects of statins: a comparative review. Expert Opin Pharmacother 2006, 7:1701–1714.
Soma MR, Corsini A, Paoletti R: Cholesterol and mevalonic acid modulation in cell metabolism and multiplication. Toxicol Lett 1992, 64–65(Spec No):1–15.
Eggens I, Eriksson LC, Chojnacki T, Dallner G: Role of dolichyl phosphate in regulation of protein glycosylation in 2-acetylaminofluorene-induced carcinogenesis in rat liver. Cancer Res 1984, 44:799–805.
Davignon J: Beneficial cardiovascular pleiotropic effects of statins. Circulation 2004, 109(23 Suppl 1):III39–43.
Hargreaves IP, Duncan AJ, Heales SJ, Land JM: The effect of HMG-CoA reductase inhibitors on coenzyme Q10: possible biochemical/clinical implications. Drug Safety 2005, 28:659–676.
Lamperti C, Naini AB, Lucchini V, et al.: Muscle coenzyme Q10 level in statin-related myopathy. Arch Neurol 2005, 62:1709–1712.
Nakamura S: Total synthesis of the squalene synthase inhibitor zaragozic acid C. Chem Pharm Bull (Tokyo) 2005, 53:1–10.
Amin D, Rutledge RZ, Needle SN, et al.: RPR 107393, a potent squalene synthase inhibitor and orally effective cholesterollowering agent: comparison with inhibitors of HMG-CoA reductase. J Pharmacol Exp Ther 1997, 281:746–752.
Sharma A, Slugg PH, Hammett JL, Jusko WJ: Clinical pharmacokinetics and pharmacodynamics of a new squalene synthase inhibitor, BMS-188494, in healthy volunteers. J Clin Pharmacol 1998, 38:1116–1121.
Hiyoshi H, Yanagimachi M, Ito M, et al.: Effect of ER-27856, a novel squalene synthase inhibitor, on plasma cholesterol in rhesus monkeys: comparison with 3-hydroxy-3-methylglutaryl-coa reductase inhibitors. J Lipid Res 2000, 41:1136–1144.
Ugawa T, Kakuta H, Moritani H, et al.: YM-53601, a novel squalene synthase inhibitor, suppresses lipogenic biosynthesis and lipid secretion in rodents. Br J Pharmacol 2003, 139:140–146.
Miki T, Kori M, Mabuchi H, et al.: Synthesis of novel 4,1-benzoxazepine derivatives as squalene synthase inhibitors and their inhibition of cholesterol synthesis. J Med Chem 2002, 45:4571–4580.
Amano Y, Nishimoto T, Tozawa R, et al.: Lipid-lowering effects of TAK-475, a squalene synthase inhibitor, in animal models of familial hypercholesterolemia. Eur J Pharmacol 2003, 466:155–161.
Nishimoto T, Tozawa R, Amano Y, et al.: Comparing myotoxic effects of squalene synthase inhibitor, T-91485, and 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors in human myocytes. Biochem Pharmacol 2003, 66:2133–2139.
Clinical Trials Database. Available at http://www.clinicaltrials.gov. Accessed September 5, 2006.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davidson, M.H. Squalene synthase inhibition: A novel target for the management of dyslipidemia. Curr Atheroscler Rep 9, 78–80 (2007). https://doi.org/10.1007/BF02693932
Issue Date:
DOI: https://doi.org/10.1007/BF02693932