Abstract
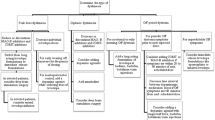
Prevention of levodopa-induced dyskinesias is a therapeutic challenge for physicians. At present, it seems only possible to delay dyskinesias and motor fluctuations. In younger patients (aged <50 years), the strategy is to use a dopamine D2 agonist as monotherapy and then to add levodopa treatment when the parkinsonian symptoms progress. In older patients, (aged >50 years to <70 years), the therapeutic approach is to use early combination therapy of levodopa and a D2 agonist.
The treatment of levodopa-induced dyskinesias must be considered in regard to the subtype and the severity of dyskinesias, and the patient. The general approach to the treatment of peak dose dyskinesias is to maintain dopamine brain stimulation at as stable a level as possible by keeping plasma and brain levodopa concentrations in the therapeutic range (above the therapeutic threshold but below the dyskinesia threshold). An appropriate strategy is to reduce the individual dose of levodopa, to spread out the daily levodopa dose and/or to try treatment with the sustained-release form of the drug. Combination treatment with the standard and sustained-release levodopa formulations is also possible. Stopping selegiline (deprenyl) therapy may reduce dyskinesias; reducing the dose of, or stopping treatment with, a dopamine agonist may also be beneficial. Anti-dyskinetic drugs such as amantadine, buspirone, fluoxetine, propanolol and principally clozapine may be used. In severe dyskinesias, apomorphine infusion may be tried. In refractory dyskinesia, surgical procedures such as pallidotomy and chronic deep brain stimulation (globus pallidus/subthalamic nucleus) may be proposed.
Theoretically, treatment of diphasic dyskinesias requires the maintenance of plasma levodopa concentrations above the dyskinesia threshold. However, this approach leads to constant and severe dyskinesia after only a few weeks of treatment. Thus, the strategy used to treat diphasic dyskinesia is close to the treatment of peak-dose dyskinesias. Apomorphine (or the liquid form of levodopa) may be helpful to prevent diphasic dyskinesias. In selected patients, a midday rest in the ‘off’ phase may decrease the duration of dyskinesia.
Treatment of early morning dystonia is based on the addition to the regimen of the sustained release formulation of levodopa before bedtime. Liquid levodopa and apomorphine injection may be used just before the appearance of the dystonic posture. Botulinum toxin may be helpful in severe dystonia.
Similar content being viewed by others
References
Marsden CD, Parkes JD, Quinn N. Fluctuations of disability in Parkinson’s disease: clinical aspects. In: Marsden CD, Fahn S, editors. Movement disorders. London: Butterworths, 1982: 96–122
Poewe WH. Clinical aspects of motor fluctuations in Parkinson’s disease. Neurology 1994; 4Suppl. 6: S6–9
Muenter MD, Sharpless NS, Tyce GM, et al. Patterns of dystonia (‘I-D-I’ and ‘D-I-D’) in response to L-dopa therapy for Parkinson’s disease. Mayo Clin Proc 1977; 52: 163–74
Lhermitte F, Agid Y, Signoret JL. Onset and end-of-dose levodopa-induced dyskinesia, possible treatment by increasing the daily doses of levodopa. Arch Neurol 1978; 35: 261–3
Poewe WH, Lees AT, Stern GM. Dystonia in Parkinsons disease: clinical and pharmacological features. Ann Neurol 1988; 23: 73–8
Koller WC, Silver DE, Lieberman A. An algorithm for the management of Parkinson’s disease. Neurology 1994; 4Suppl. 10: S1–52
Marconi R, Lefebvre-Caparros D, Bonnet AM, et al. Levodopa-induced dyskinesia in Parkinson’s disease: phenomenology and pathophysiology. Mov Disord 1994; 9: 2–12
Vidailhet M, Bonnet AM, Marconi R, et al. Do parkinsonian symptoms and levodopa-induced dyskinesias start in the foot? Neurology 1994; 44: 1613–6
Nutt JG. Levodopa-induced dyskinesia: review, observations and speculations. Neurology 1990; 40: 340–5
Poewe WH, Lees AJ, Stern GM. Low-dose L-dopatherapy in Parkinsons disease: a 6-years follow-up study. Neurology 1986; 36: 1528–30
Mouradian MM, Chase TN. Improved dopaminergic therapy of Parkinsons disease. In: Marsden CD, Fahn S, editors. Movement disorders 3. London: Butterworths, 1994: 180–99
Nutt JG. Pharmacokinetics and pharmacodynamics of levodopa: clinical implications. In: Koller WC, editor. Handbook of Parkinson’s disease. 2nd rev. ed. New York: Marcel Dekker Inc., 1992: 411–31
Mouradian MM, Heuser IJE, Baronti F, et al. Pathogenesis of dyskinesias in Parkinson’s disease. Ann Neurol 1989; 25: 523–6
Lees AJ, Stern GM. Sustained bromocriptine therapy in previously untreated patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry 1981; 44: 1020–3
Montastruc JL, Rascol O, Sennard JM, et al. Arandomized control study of bromocriptine versus levodopa previously untreated parkinsonian patients: a 3-years follow-up. J Neurol Neurosurg Psychiatry 1989; 52: 773–5
Kostic V, Przedborski S, Flaster E, et al. Early development of levodopa-induced dyskinesias and response fluctuations in young-onset Parkinsons disease. Neurology 1991; 41: 202–5
Schuh LA, Bennett JP. Suppression of dyskinesias in Parkinson’s disease: continuous intravenous levodopa shifts the dose-response for production of dyskinesias but not relief of parkinsonism in patients with advanced Parkinson’s disease. Neurology 1993; 43: 1545–50
Dupont E, Andersen A, Boas J, et al. Sustained-release madopar HBS registered compared with standard madopar registered in the long-term treatment of de novo parkinsonian patients. Acta Neurol Scand 1996; 93: 14–20
Block G, Liss C, Reines S, et al. Comparison of immediate-release and controlled release carbidopa/levodopa in Parkinson’s disease. Eur Neurol 1997; 37: 23–7
Rinne UK. Combined bromocriptine-levodopa therapy early in Parkinsons disease. Neurology 1985, 35: 1196–8
Weiner WJ, Factor JA, Sanchez-Ramoz JR, et al. Early combination therapy (bromocriptine and levodopa) does not prevent motor fluctuations in Parkinsons disease. Neurology 1993; 43: 21–6
Gimenez-Roldan S, Tolosa E, Burguera JA, et al. Early combination of bromocriptine and levodopa in Parkinsons disease: a propective randomized study of two parallel groups over a total follow-up period of 44 months including an initial 8 months double blind stage. Clin Neuropharmacol 1997; 20: 67–76
Quinn N. Drug treatment of Parkinsons disease. BMJ 1995; 310: 575–9
Parkinsons Study Group. Effects of deprenyl on the progression of disability in early Parkinsons disease. N Engl J Med 1989; 321: 1364–71
Parkinsons Study Group. Effects of tocopherol and deprenyl on the progression of disability in early Parkinsons disease. N Engl J Med 1993; 328: 176–83
Yahr MD, Elizan TS, Mores D. Selegiline in the treatment of Parkinsons disease: long-term experience. Acta Neurol Scand 1989; 126: 157–61
Brannan T, Yahr MD. Comparative study of selegiline plus L-dopa-carbidopa versus L-dopa-carbidopa alone is the treatment of Parkinsons disease. Ann Neurol 1995; 37: 95–8
Pahwa R, Busenbark K, Uber SJ. Clinical experience with controlled-release levodopa/carbidopa in Parkinsons disease. Neurology 1993; 43: 677–81
Rodnitzky RL. The use of sinemet CR in the management of mild to moderate Parkinsons disease. Neurology 1992; 42Suppl. 1: 44–50
Verhagen Metman L, Del Dotto P, Van den Munckhof P, et al. Amantadine as treatment for dyskinesias and motor fluctuations in Parkinson’s disease. Neurology 1998; 50: 1323–6
Bonifati V, Fabrizio E, Cipriani R, et al. Buspirone in levodopa-induced dyskinesias. Clin Neuropharmacol 1994; 17: 73–82
Durif F, Vidailhet M, Bonnet AM, et al. Levodopa-induced dyskinesia are improved by fluoxetine. Neurology 1995; 45: 1855–8
Carpentier AF, Bonnet AM, Vidailhet M, et al. Improvement of levodopa induced dyskinesias by propanolol in Parkinson’s disease. Neurology 1996; 46: 1548–51
Bennett JP, Landow ER, Dietrich S, et al. Suppression of dyskinesias in advanced Parkinson’s disease: moderate daily clozapine doses provide long-term dyskinesia reduction. Mov Disord 1994; 9: 409–14
Durif F, Vidailhet M, Assal F, et al. Low dose clozapine improves dyskinesias in Parkinson’s disease. Neurology 1997; 48: 658–61
Hughes AJ, Bishop S, Kleedorfer B, et al. Subcutaneous apomorphine in Parkinson’s disease: response to chronic administration for five years. Mov Disord 1993; 8: 165–70
Baron S, Vitek JL, Bakay RAE, et al. Treatment of advanced Parkinsons disease by posterior GPI pallidotomy: one year results of a pilot study. Ann Neurol 1996; 40: 355–66
Pahwa R, Wilkinson S, Smith D, et al. High frequency stimulation of the globus pallidus for the treatment of the Parkinsons disease. Neurology 1997; 49: 249–53
Krack P, Limousin P, Benabid AL, et al. Chronic stimulation of subthalamic nucleus improves levodopa-induced dyskinesias in Parkinsons disease. Lancet 1998; 350: 1676
Fahn S. Fluctuations of disabiliting in Parkinsons disease: pathophysiology. In: Marsden CD, Fahn S, editors. Movement disorders. London: Butterworth, 1982: 123–45
Facca A, Sanchez-Ramos J. High dose pergolide monotherapy in the treatment of severe levodopa-induced dyskinesias. Mov Disord 1996; 11: 327–9
De Saint Victor JF, Pollak P, Gervason CN, et al. Levodopa-induced diphasic dyskinesias improved by subcutaneous apomorphine. Mov Disord 1992; 3: 283
Durif F, Deffond D, Dordain G, et al. Apomorphine and diphasic dyskinesia. Clin Neuropharmacol 1994; 17: 99–102
Pourcher E, Bonnet AM, Kefalos J, et al. Effects of etybenzatropine and diazepam on levodopa induced diphasic dyskinesias in Parkinson’s disease. Mov Disord 1989; 4: 195–201
Pacchetti C, Albani G, Martignoni E, et al. Off painful dystonia in Parkinsons disease treated with botulinum toxin. Mov Disord 1995; 10: 333–6
Baronti F, Mouradian MM, Connan KE, et al. A partial dopamine agonist therapy of levodopa-induced dyskinesias. Neurology 1992; 42: 1241–3
Brefel C, Blin O, Descombes S, et al. ABT-431, a selective D1 agonist has efficacy in patients with Parkinson’s Disease. XII International Symposium on Parkinson’s Disease; 1997 Mar 23–26; London
Rascol O, Arnulf I, Drefel C, et al. L-dopa induced dyskinesias improvement by an alpha 2 antagonist, idazoxan in patients with Parkinson’s Disease. XII International Symposium on Parkinsondisease: 1997 Mar 23–26; London
Papa SM, Gees TN. Levodopa-induced dyskinesias improved by glutamate antagonist in parkinsonian monkeys. Ann Neurol 1996; 39: 574–8
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Durif, F. Treating and Preventing Levodopa-Induced Dyskinesias. Drugs Aging 14, 337–345 (1999). https://doi.org/10.2165/00002512-199914050-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-199914050-00002