Abstract
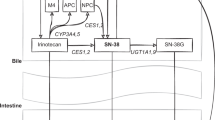
The metabolism of irinotecan (CPT-11) involves sequential activation to SN-38 and detoxification to the pharmacologically inactive SN-38 glucuronide (SN-38G). We have previously demonstrated the role of UGT1A1 enzyme in the glucuronidation of SN-38 and a significant correlation between in vitro glucuronidation of SN-38 and UGT1A1 gene promoter polymorphism. This polymorphism (UGT1A1*28) is characterized by the presence of an additional TA repeat in the TATA sequence of the UGT1A1 promoter, ((TA)7TAA, instead of (TA)6TAA). Here we report the results from a prospective clinical pharmacogenetic study to determine the significance of UGT1A1*28 polymorphism on irinotecan disposition and toxicity in patients with cancer. Twenty patients with solid tumors were treated with a 90 min i.v. infusion of irinotecan (300 mg m−2) once every 3 weeks. The frequency of UGT1A1 genotypes was as follows: 6/6—45%, 6/7—35% and 7/7—20%, with allele frequencies of 0.375 and 0.625 for (TA)7TAA and (TA)6TAA, respectively. Patients with the (TA)7TAA polymorphism had significantly lower SN-38 glucuronidation rates than those with the normal allele (6/6>6/7>7/7, P = 0.001). More severe grades of diarrhea and neutropenia were observed only in patients heterozygous (grade 4 diarrhea, n = 1) or homozygous (grade 3 diarrhea/grade 4 neutropenia, n = 1 and grade 3 neutropenia, n = 1) for the (TA)7TAA sequence. The results suggest that screening for UGT1A1*28 polymorphism may identify patients with lower SN-38 glucuronidation rates and greater susceptibility to irinotecan induced gastrointestinal and bone marrow toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Saltz LB, Cox JV, Blanke C, Rosen LS, Fehrenbacher L, Moore MJ et al . Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group N Engl J Med 2000 343: 905–914
Iyer L, King CD, Whitington PF, Green MD, Roy SK, Tephly TR et al . Genetic predisposition to the metabolism of irinotecan (CPT-11). Role of uridine diphosphate glucuronosyltransferase isoform 1A1 in the glucuronidation of its active metabolite (SN-38) in human liver microsomes J Clin Invest 1998 101: 847–854
Gupta E, Lestingi TM, Mick R, Ramirez J, Vokes EE, Ratain MJ . Metabolic fate of irinotecan in humans: correlation of glucuronidation with diarrhea Cancer Res 1994 54: 3723–3725
Crigler JF, Najjar . Congenital familial nonhemolytic jaundice with kernicterus Pediatrics 1952 10: 169–180
Bosma PJ, Chowdhury JR, Bakker C, Gantla S, Boer A, Oostra BA . The genetic basis of the reduced expression of bilirubin UDP-glucuronosyltransferase 1 in Gilbert's syndrome N Engl J Med 1995 333: 1171–1175
Iyer L, Hall D, Das S, Mortell MA, Ramirez J, Kim S et al . Phenotype-genotype correlation of in vitro SN-38 (active metabolite of irinotecan) and bilirubin glucuronidation in human liver tissue with UGT1A1 promoter polymorphism Clin Pharmacol Ther 1999 65: 576–582
Beutler E, Gelbart T, Demina A . Racial variability in the UDP-glucuronosyltransferase 1 (UGT1A1) promoter: a balanced polymorphism for regulation of bilirubin metabolism? Proc Natl Acad Sci U S A 1998 95: 8170–8174
Monaghan G, Ryan M, Seddon R, Hume R, Burchell B . Genetic variation in bilirubin UPD-glucuronosyltransferase gene promoter and Gilbert's syndrome Lancet 1996 347: 578–581
Hall D, Ybazeta G, Destro-Bisol G, Petzl-Erler ML, Di Rienzo A . Variability at the uridine diphosphate glucuronosyltransferase 1A1 promoter in human populations and primates Pharmacogenetics 1999 9: 591–599
Takasuna K, Kasai Y, Kitano Y, Mori K, Kakihata K, Hirohashi M et al . Study on the mechanisms of diarrhea induced by the new anticancer drug irinotecan hydrochloride (CPT-11) Folia Pharmacol Jpn 1995 105: 447–460
Gupta E, Mick R, Ramirez J, Wang X, Lestingi TM, Vokes EE et al . Pharmacokinetic and pharmacodynamic evaluation of the topoisomerase inhibitor irinotecan in cancer patients J Clin Oncol 1997 15: 1502–1510
Wasserman E, Myara A, Lokiec F, Goldwasser F, Trivin F, Mahjoubi M . Severe CPT-11 toxicity in patients with Gilbert's syndrome: two case reports Ann Oncol 1997 8: 1049–1051
Ando Y, Saka H, Asai G, Sugiura S, Shimokata K, Kamataki T . UGT1A1 genotypes and glucuronidation of SN-38, the active metabolite of irinotecan Annals Oncol 1998 9: 845–847
Ando Y, Saka H, Ando M, Sawa T, Muro K, Ueoka H et al . Polymorphisms of UDP-glucuronosyltransferase gene and irinotecan toxicity: a pharmacogenetic analysis Cancer Res 2000 60: 6921–6926
Sargent DJ, Niedzwiecki D, O'Connell MJ, Schilsky RL . Recommendation for caution with irinotecan, fluorouracil, and leucovorin for colorectal cancer (letter) New Eng J Med 2001 345: 144–145
Huang CS, Luo GA, Huang ML, Yu SC, Yang SS . Variations of the bilirubin uridine-diphosphoglucuronosyl transferase 1A1 gene in healthy Taiwanese Pharmacogenetics 2000 10: 539–544
Akaba K, Kimura T, Sasaki A, Tanabe S, Wakabayashi T, Hiroi M et al . Neonatal hyperbilirubinemia and a common mutation of the bilirubin uridine diphosphate-glucuronosyltransferase gene in Japanese J Hum Genet 1 999 44: 22–25
Akaba K, Kimura T, Sasaki A, Tanabe S, Ikegami T, Hashimoto M et al . Neonatal hyperbilirubinemia and mutation of the bilirubin uridine diphosphate-glucuronosyltransferase gene: a common missense mutation among Japanese, Koreans and Chinese Biochem Mol Biol Int 1998 46: 21–26
Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J et al . Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression Nat Genet 2001 27: 383–391
Hoffmeyer S, Burk O, von Richter O, Arnold HP, Brockmoller J, Johne A et al . Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo Proc Natl Acad Sci U S A 2000 97: 3473–3478
Iyer L, Ramirez J, Shepard DR, Hossfeld D-K, Ratain MJ, Mayer U . Role of p-glycoprotein (Pgp) in biliary excretion of irinotecan (CPT-11) in normal and mdr1a/1b(−/−) mice Drug Metab Rev 2000 32: 196 (Abst)
Pitot HC, Goldberg RM, Reid JM, Sloan JA, Skaff PA, Erlichman C et al . Phase I dose-finding and pharmacokinetic trial of irinotecan hydrochloride (CPT-11) using a once-every-three-week dosing schedule for patients with advanced solid tumor malignancy Clin Cancer Res 2000 6: 2236–2244
Cuzick J . A Wilcoxon-type test for trend Statistics in Medicine 1985 4: 87–90
Acknowledgements
Supported by grants GM61393, CA69852–07 and RR00055.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iyer, L., Das, S., Janisch, L. et al. UGT1A1*28 polymorphism as a determinant of irinotecan disposition and toxicity. Pharmacogenomics J 2, 43–47 (2002). https://doi.org/10.1038/sj.tpj.6500072
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.tpj.6500072
Keywords
This article is cited by
-
Integration of DNA sequencing with population pharmacokinetics to improve the prediction of irinotecan exposure in cancer patients
British Journal of Cancer (2022)
-
Prediction of Drug–Drug Interaction Between Dabrafenib and Irinotecan via UGT1A1-Mediated Glucuronidation
European Journal of Drug Metabolism and Pharmacokinetics (2022)
-
A dose regimen-finding study to evaluate the safety, tolerability, pharmacokinetics, and activity of oratecan in subjects with advanced malignancies
Cancer Chemotherapy and Pharmacology (2022)
-
The Underestimated and Overlooked Burden of Diarrhea and Constipation in Cancer Patients
Current Oncology Reports (2022)
-
Epigenetics and microRNAs in UGT1As
Human Genomics (2021)