Abstract
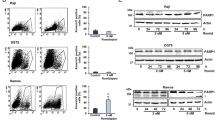
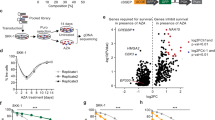
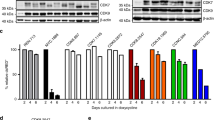
Interactions between the histone deacetylase inhibitor SAHA and the pharmacologic MEK1/2 inhibitor PD184352 were examined in Bcr/Abl+ human leukemia cells. Coadministration of minimally toxic concentrations of SAHA (or sodium butyrate) and PD184352 (or U0126) resulted in a synergistic increase in mitochondrial damage, caspase activation, and apoptosis in K562 and LAMA 84 cells. Similar interactions were observed in CD34+ cells from two patients with CML and in imatinib mesylate-resistant K562 cells but not in normal human CD34+ bone marrow cells. These events were associated with a marked increase in ROS generation, inactivation of ERK and Akt, downregulation of p21CIP1, Bcr/Abl, and cyclin D1, and activation of JNK. Of these events, ROS generation, ERK inactivation, and cytochrome c/AIF release were largely caspase-independent, whereas the other phenomena displayed varying degrees of caspase-dependence. Using pharmacologic and genetic approaches, generation of ROS, p21CIP1 downregulation, and inactivation of Akt and MEK were found to play significant functional roles in SAHA/PD184352-mediated lethality, whereas JNK activation and Raf-1 downregulation were determined to represent secondary events. These findings indicate that interruption of the MEK/ERK pathway substantially lowers the threshold for HDAC inhibitor-mediated oxidative injury, mitochondrial dysfunction, and apoptosis, suggesting that this approach warrants further examination in Bcr/Abl+-related malignancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Faderl S, Talpaz M, Estrov Z, O'Brien S, Kurzrock R, Kantarjian HM . The biology of chronic myeloid leukemia. N Engl J Med 1999; 341: 164–172.
Horita M, Andreu EJ, Benito A, Arbona C, Sanz C, Benet I et al. Blockade of the Bcr-Abl kinase activity induces apoptosis of chronic myelogenous leukemia cells by suppressing signal transducer and activator of transcription 5-dependent expression of Bcl-xL. J Exp Med 2000; 191: 977–984.
Hochhaus A, Kreil S, Corbin AS, La Rosee P, Muller MC, Lahaye T et al. Molecular and chromosomal mechanisms of resistance to imatinib (STI571) therapy. Leukemia 2000; 16: 2190–2196.
Donato NJ, Wu JY, Stapley J, Lin H, Arlinghaus R, Aggarwal BB et al. Imatinib mesylate resistance through BCR-ABL independence in chronic myelogenous leukemia. Cancer Res 2004; 64: 672–677.
Van Etten RA . Mechanisms of transformation by the BCR-ABL oncogene: new perspectives in the post-imatinib era. Leuk Res 2004; 28 (Suppl 1): S21–S28.
Bedi A, Barber JP, Bedi GC, el-Deiry WS, Sidransky D, Vala MS et al. BCR-ABL-mediated inhibition of apoptosis with delay of G2/M transition after DNA damage: a mechanism of resistance to multiple anticancer agents. Blood 1995; 86: 1148–1158.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 1996; 2: 561–566.
Knight GW, McLellan D . Use and limitations of imatinib mesylate (Glivec), a selective inhibitor of the tyrosine kinase Abl transcript in the treatment of chronic myeloid leukaemia. Br J Biomed Sci 2004; 61: 103–111.
von Bubnoff N, Peschel C, Duyster J . Resistance of Philadelphia-chromosome positive leukemia towards the kinase inhibitor imatinib (STI571, Glivec): a targeted oncoprotein strikes back. Leukemia 2003; 17: 829–838.
Shah NP, Tran C, Lee FY, Chen P, Norris D, Sawyers CL . Overriding imatinib resistance with a novel ABL kinase inhibitor. Science 2004; 305: 399–401.
Nakatani Y . Histone acetylases – versatile players. Genes Cells 2001; 6: 79–86.
Johnstone RW . Histone-deacetylase inhibitors: novel drugs for the treatment of cancer. Nat Rev Drug Discov 2002; 1: 287–299.
Dai Y, Rahmani M, Grant S . An intact NF-kappaB pathway is required for histone deacetylase inhibitor-induced G1 arrest and maturation in U937 human myeloid leukemia cells. Cell Cycle 2003; 2: 467–472.
Richon VM, Webb Y, Merger R, Sheppard T, Jursic B, Ngo L et al. Second generation hybrid polar compounds are potent inducers of transformed cell differentiation. Proc Natl Acad Sci USA 1996; 93: 5705–5708.
Vrana JA, Decker RH, Johnson CR, Wang Z, Jarvis WD, Richon VM et al. Induction of apoptosis in U937 human leukemia cells by suberoylanilide hydroxamic acid (SAHA) proceeds through pathways that are regulated by Bcl-2/Bcl-XL, c-Jun, and p21CIP1, but independent of p53. Oncogene 1999; 18: 7016–7025.
Ruefli AA, Ausserlechner MJ, Bernhard D, Sutton VR, Tainton KM, Kofler R et al. The histone deacetylase inhibitor and chemotherapeutic agent suberoylanilide hydroxamic acid (SAHA) induces a cell-death pathway characterized by cleavage of Bid and production of reactive oxygen species. Proc Natl Acad Sci USA 2001; 98: 10833–10838.
George P, Bali P, Annavarapu S, Scuto A, Fiskus W, Guo F et al. Combination of the histone deacetylase inhibitor LBH589 and the hsp90 inhibitor 17-AAG is highly active against human CML-BC cells and AML cells with activating mutation of FLT-3. Blood 2005; 105: 1768–1776.
Yu X, Guo ZS, Marcu MG, Neckers L, Nguyen DM, Chen GA et al. Modulation of p53, ErbB1, ErbB2, and Raf-1 expression in lung cancer cells by depsipeptide FR901228. J Natl Cancer Inst 2002; 94: 504–513.
Insinga A, Monestiroli S, Ronzoni S, Gelmetti V, Marchesi F, Viale A et al. Inhibitors of histone deacetylases induce tumor-selective apoptosis through activation of the death receptor pathway. Nat Med 2005; 11: 71–76.
Maggio SC, Rosato RR, Kramer LB, Dai Y, Rahmani M, Paik DS et al. The histone deacetylase inhibitor MS-275 interacts synergistically with fludarabine to induce apoptosis in human leukemia cells. Cancer Res 2004; 64: 2590–2600.
Yu C, Subler M, Rahmani M, Reese E, Krystal G, Conrad D et al. Induction of apoptosis in Bcr/Abl+ cells by histone deacetylase inhibitors involves reciprocal effects on the RAF-11/MEK/ERK and JNK pathways. Cancer Biol Ther 2003; 2: 544–551.
Minden A, Karin M . Regulation and function of the JNK subgroup of MAP kinases. Biochim Biophys Acta 1997; 1333: F85–F104.
Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME . Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 1995; 270: 1326–1331.
Wang X, Martindale JL, Liu Y, Holbrook NJ . The cellular response to oxidative stress: influences of mitogen-activated protein kinase signalling pathways on cell survival. Biochem J 1998; 333 (t2): 291–300.
Sebolt-Leopold JS . Development of anticancer drugs targeting the MAP kinase pathway. Oncogene 2000; 19: 6594–6599.
Allen LF, Sebolt-Leopold J, Meyer MB . CI-1040 (PD184352), a targeted signal transduction inhibitor of MEK (MAPKK). Semin Oncol 2003; 30 (Suppl 16): 105–106.
Dai Y, Yu C, Singh V, Tang L, Wang Z, McInistry R et al. Pharmacological inhibitors of the mitogen-activated protein kinase (MAPK) kinase/MAPK cascade interact synergistically with UCN-01 to induce mitochondrial dysfunction and apoptosis in human leukemia cells. Cancer Res 2001; 61: 5106–5115.
Dai Y, Rahmani M, Pei XY, Khanna P, Han SI, Mitchell C et al. Farnesyltransferase inhibitors interact synergistically with the CHK1 inhibitor UCN-01 to induce apoptosis in human leukemia cells through interruption of both AKT and MEK/ERK pathways and activation of SEK1/JNK. Blood 2005; 105: 1706–1716.
Philpott NJ, Turner AJ, Scopes J, Westby M, Marsh JC, Gordon-Smith EC et al. The use of 7-amino actinomycin D in identifying apoptosis: simplicity of use and broad spectrum of application compared with other techniques. Blood 1996; 87: 2244–2251.
Chou TC, Talalay P . Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 1984; 22: 27–55.
Dai Y, Rahmani M, Corey SJ, Dent P, Grant S . A Bcr/Abl-independent, Lyn-dependent form of imatinib mesylate (STI-571) resistance is associated with altered expression of Bcl-2. J Biol Chem 2004; 279: 34227–34239.
Almenara J, Rosato R, Grant S . Synergistic induction of mitochondrial damage and apoptosis in human leukemia cells by flavopiridol and the histone deacetylase inhibitor suberoylanilide hydroxamic acid (SAHA). Leukemia 2002; 16: 1331–1343.
Liu Y, Martindale JL, Gorospe M, Holbrook NJ . Regulation of p21WAF1/CIP1 expression through mitogen-activated protein kinase signaling pathway. Cancer Res 1996; 56: 31–35.
Rahmani M, Yu C, Dai Y, Reese E, Ahmed W, Dent P et al. Coadministration of the heat shock protein 90 antagonist 17-allylamino-17-demethoxygeldanamycin with suberoylanilide hydroxamic acid or sodium butyrate synergistically induces apoptosis in human leukemia cells. Cancer Res 2003; 63: 8420–8427.
Butler LM, Zhou X, Xu WS, Scher HI, Rifkind RA, Marks PA et al. The histone deacetylase inhibitor SAHA arrests cancer cell growth, up-regulates thioredoxin-binding protein-2, and down-regulates thioredoxin. Proc Natl Acad Sci USA 2002; 99: 11700–11705.
Ungerstedt JS, Sowa Y, Xu WS, Shao Y, Dokmanovic M, Perez G et al. Role of thioredoxin in the response of normal and transformed cells to histone deacetylase inhibitors. Proc Natl Acad Sci USA 2005; 102: 673–678.
Rossig L, Badorff C, Holzmann Y, Zeiher AM, Dimmeler S . Glycogen synthase kinase-3 couples AKT-dependent signaling to the regulation of p21Cip1 degradation. J Biol Chem 2002; 277: 9684–9689.
Kondapaka SB, Singh SS, Dasmahapatra GP, Sausville EA, Roy KK . Perifosine, a novel alkylphospholipid, inhibits protein kinase B activation. Mol Cancer Ther 2003; 2: 1093–1103.
Widmann C, Gibson S, Johnson GL . Caspase-dependent cleavage of signaling proteins during apoptosis. A turn-off mechanism for anti-apoptotic signals. J Biol Chem 1998; 273: 7141–7147.
Dasmahapatra GP, Didolkar P, Alley MC, Ghosh S, Sausville EA, Roy KK . In vitro combination treatment with perifosine and UCN-01 demonstrates synergism against prostate (PC-3) and lung (A549) epithelial adenocarcinoma cell lines. Clin Cancer Res 2004; 10: 5242–5252.
Bachelder RE, Wendt MA, Fujita N, Tsuruo T, Mercurio AM . The cleavage of Akt/protein kinase B by death receptor signaling is an important event in detachment-induced apoptosis. J Biol Chem 2001; 276: 34702–34707.
Wang X, McCullough KD, Franke TF, Holbrook NJ . Epidermal growth factor receptor-dependent Akt activation by oxidative stress enhances cell survival. J Biol Chem 2000; 275: 14624–14631.
Shelton JG, Blalock WL, White ER, Steelman LS, McCubrey JA . Ability of the activated PI3K/Akt oncoproteins to synergize with MEK1 and induce cell cycle progression and abrogate the cytokine-dependence of hematopoietic cells. Cell Cycle 2004; 3: 503–512.
Neshat MS, Raitano AB, Wang HG, Reed JC, Sawyers CL . The survival function of the Bcr-Abl oncogene is mediated by Bad-dependent and -independent pathways: roles for phosphatidylinositol 3-kinase and Raf. Mol Cell Biol 2000; 20: 1179–1186.
Hochhaus A, Kreil S, Corbin AS et al. Molecular and chromosomal mechanisms of resistance to imatinib (STI571) therapy. Leukemia 2002; 16: 2190–2196.
Shah NP, Nicoll JM, Nagar B, Gorre ME, Paquette RL, Kuriyan J et al. Multiple BCR-ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2002; 2: 117–125.
Acknowledgements
This work was supported by awards CA63753, CA93738, and CA 100866, CA88906, CA72955 from the NIH, A Translational Research Award from the Leukemia and Lymphoma Society of America (6045-03), an award from the Department of Defense (DAMD-17-03-1-0209), an award from the V Foundation, and by the Universal Professorship (to PD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu).
Rights and permissions
About this article
Cite this article
Yu, C., Dasmahapatra, G., Dent, P. et al. Synergistic interactions between MEK1/2 and histone deacetylase inhibitors in BCR/ABL+ human leukemia cells. Leukemia 19, 1579–1589 (2005). https://doi.org/10.1038/sj.leu.2403868
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403868
Keywords
This article is cited by
-
Integrated Drug Expression Analysis for leukemia: an integrated in silico and in vivo approach to drug discovery
The Pharmacogenomics Journal (2017)
-
MEK inhibitors as a chemotherapeutic intervention in multiple myeloma
Blood Cancer Journal (2013)
-
Preclinical evaluation of dual PI3K-mTOR inhibitors and histone deacetylase inhibitors in head and neck squamous cell carcinoma
British Journal of Cancer (2012)
-
Synergistic effect of bortezomib and valproic acid treatment on the proliferation and apoptosis of acute myeloid leukemia and myelodysplastic syndrome cells
Annals of Hematology (2011)
-
Activating Transcription Factor 3 regulates in part the enhanced tumour cell cytotoxicity of the histone deacetylase inhibitor M344 and cisplatin in combination
Cancer Cell International (2010)