Abstract
Background Many patients with coronary heart disease do not achieve recommended LDL-cholesterol levels, due to either intolerance or inadequate response to available lipid-lowering therapy. Microsomal triglyceride transfer protein (MTP) inhibitors might provide an alternative way to lower LDL-cholesterol levels. We tested the safety and LDL-cholesterol-lowering efficacy of an MTP inhibitor, AEGR-733 (Aegerion Pharmaceuticals Inc., Bridgewater, NJ), alone and in combination with ezetimibe.
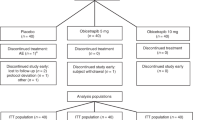
Methods We performed a multicenter, double-blind, 12-week trial, which included 84 patients with hypercholesterolemia. Patients were randomly assigned ezetimibe 10 mg daily (n = 29); AEGR-733 5.0 mg daily for the first 4 weeks, 7.5 mg daily for the second 4 weeks and 10 mg daily for the last 4 weeks (n = 28); or ezetimibe 10 mg daily and AEGR-733 administered with the dose titration described above (n = 28).
Results Ezetimibe monotherapy led to a 20–22% decrease in LDL-cholesterol concentrations. AEGR-733 monotherapy led to a dose-dependent decrease in LDL-cholesterol concentration: 19% at 5.0 mg, 26% at 7.5 mg and 30% at 10 mg. Combined therapy produced similar but larger dose-dependent decreases (35%, 38% and 46%, respectively). The number of patients who discontinued study drugs owing to adverse events was five with ezetimibe alone, nine with AEGR-733 alone, and four with combined ezetimibe and AEGR-733. Discontinuations from AEGR-733 were due primarily to mild transaminase elevations.
Conclusions Inhibition of LDL production with low-dose AEGR-733, either alone or in combination with ezetimibe, could be an effective therapeutic option for patients unable to reach target LDL-cholesterol levels.
Key Points
-
Many patients with coronary heart disease cannot achieve current target levels for LDL-cholesterol owing to either intolerance or inadequate response to conventional lipid-lowering therapy; new treatment strategies are required
-
A possible therapeutic approach is inhibition of microsomal triglyceride transfer protein, which is essential for the assembly and secretion of apolipoprotein-B-containing lipoproteins
-
AEGR-733, alone and in combination with ezetimibe, had notable LDL-cholesterol-lowering effects in patients with hyperlipidemia
-
The main side effect associated with AEGR-733 was elevation in transaminase concentrations, which returned to baseline values after cessation of therapy; gastrointestinal side effects were minor
-
Low-dose microsomal triglyceride transfer protein inhibitors, alone or in combination with ezetimibe, could be an effective therapeutic option for patients unable to reach target LDL levels with conventional therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (2001) Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 285: 2486–2497
Grundy SM et al. (2004) Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. Circulation 110: 227–239
Smith SC Jr et al. (2006) AHA/ACC Guidelines for Secondary Prevention for Patients With Coronary and Other Atherosclerotic Vascular Disease: 2006 Update: endorsed by the National Heart, Lung, and Blood Institute. Circulation 113: 2363–2372
Ballantyne CM et al. (2005) Dose-comparison study of the combination of ezetimibe and simvastatin (Vytorin) versus atorvastatin in patients with hypercholesterolemia: the Vytorin Versus Atorvastatin (VYVA) Study. Am. Heart J 149: 464–473
Baigent C et al. (2005) Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 366: 1267–1278
Andrews T et al. (2001) Achieving and maintaining national cholesterol education program low-density lipoprotein cholesterol goals with five Statins. Am J Med 111: 185–191
Alsheikh-Ali AA and Karas RH (2005) Adverse events with concomitant amiodarone and statin therapy. Prev Cardiol 8: 95–97
Ballantyne CM et al. (2003) Risk for myopathy with statin therapy in high-risk patients. Arch Intern Med 163: 553–564
Burnett JR and Watts GF (2007) MTP inhibition as a treatment for dyslipidaemias: time to deliver or empty promises. Expert Opin Ther Targets 11: 181–189
Sharp D et al. (1993) Cloning and gene defects in microsomal triglyceride transfer protein associated with abetalipoproteinaemia. Nature 365: 65–69
Rader DJ and Brewer HB (1993) Abetalipoproteinemia. New insights into lipoprotein assembly and vitamin E metabolism from a rare genetic disease. JAMA 270: 865–869
Liao W et al. (2003) Blocking microsomal triglyceride transfer protein interferes with apoB secretion without causing retention or stress in the ER. J Lipid Res 44: 978–985
Wetterau JR et al. (1998) An MTP inhibitor that normalizes atherogenic lipoprotein levels in WHHL rabbits. Science 282: 751–754
Cuchel M et al. (2007) Inhibition of microsomal triglyceride transfer protein in familial hypercholesterolemia. N Engl J Med 356: 148–156
Svedlund J et al. (1988) GSRS—A clinical rating scale for gastrointestinal symptoms in patients with irritable bowel syndrome and peptic ulcer disease. Dig Dis Sci 33: 129–134
Revicki DA et al. (1998) Reliability and validity of the Gastrointestinal Symptom Rating Scale in patients with gastroesophageal reflux disease. Qual Life Res 1: 75–83
Kulich KR et al. (2003) Psychometric validation of the German translation of the Gastrointestinal rating scale (GSRS) and Quality of Life in Reflux and Dyspepsia (QUOLRAD) Questionnaire in patients with reflux disease. Health Qual Life Outcomes 7: 62
Davidson MH and Robinson JG (2006) Lipid-lowering effects of statins: a comparative review. Expert Opin Pharmacother 7: 1701–1714
Knopp RH et al. (2003) Effects of ezetimibe, a new cholesterol absorption inhibitor, on plasma lipids in patients with primary hypercholesterolemia. Eur Heart J 24: 729–741
Chandler CE et al. (2003) CP-346086: an MTP inhibitor that lowers plasma total, VLDL, and LDL cholesterol and triglycerides by up to 70% in experimental animals and in humans. J Lipid Res 44: 1887–1901
Stern L et al. (2004) The effects of low-carbohydrate versus conventional weight loss diets in severely obese adults: one-year follow-up of a randomized trial. Ann Intern Med 140: 778–785
Ornish D et al. (1998) Intensive lifestyle changes for reversal of coronary heart disease. JAMA 280: 2001–2007
Ikewaki K et al. (1994) In vivo metabolism of apolipoproteins A-I and E in patients with abetalipoproteinemia: implications for the roles of apolipoproteins B and E in HDL metabolism. J Lipid Res 35: 1809–1819
Guerciolini R et al. (2001) comparative evaluation of fecal fat excretion induced by orlistat and chitosan. Obesity Res 9: 364–367
Temel RE et al. (2007) Hepatic Niemann-Pick C1–like 1 regulates biliary cholesterol concentration and is a target of ezetimibe. J Clin Invest 117: 1968–1978.
Sorbera LA et al. (2000) Implitapide. Drugs Fut 25: 1138–1144 [20002511]
Zaiss S et al. (1999) BAY 13-9952, an inhibitor of the microsomal triglyceride transfer protein (MTP), inhibits atherosclerotic plaque formation in cholesterol-fed rabbits in spite of a concomitant reductio of vitamin E [Abstr]. Circulation. 100 (Suppl 1): 258
Zaiss S and Sander E (2000) Bay 13-9952 (implitapide), an inhibitor of the microsomal triglyceride transfer protein (MTP), inhibits atherosclerosis and prolongs lifetime in apoE knockout mice. Eur Heart J 21 (Suppl): 16
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This study was funded by Aegerion Inc.
FF Samaha Samaha is funded by Abbott laboratories, Aegerion Inc., and Merck Inc.
J McKenney provides consulting services for Abbott Laboratories, Aegerion Inc., AstraZeneca, Daiichi Sankyo and Merck Inc.
WJ Sasiela is an employee of Aegerion Inc.
DJ Rader is a stockholder and member of the scientific advisory board in Aegerion Inc.
Rights and permissions
About this article
Cite this article
Samaha, F., McKenney, J., Bloedon, L. et al. Inhibition of microsomal triglyceride transfer protein alone or with ezetimibe in patients with moderate hypercholesterolemia. Nat Rev Cardiol 5, 497–505 (2008). https://doi.org/10.1038/ncpcardio1250
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio1250
This article is cited by
-
Lipid management in patients with chronic kidney disease
Nature Reviews Nephrology (2018)
-
Efficacy and Safety of Lomitapide in Hypercholesterolemia
American Journal of Cardiovascular Drugs (2017)
-
Impact of L-carnitine on plasma lipoprotein(a) concentrations: A systematic review and meta-analysis of randomized controlled trials
Scientific Reports (2016)
-
Future Directions to Establish Lipoprotein(a) as a Treatment for Atherosclerotic Cardiovascular Disease
Cardiovascular Drugs and Therapy (2016)
-
Emerging Therapeutic Options for Lowering of Lipoprotein(a): Implications for Prevention of Cardiovascular Disease
Current Atherosclerosis Reports (2016)