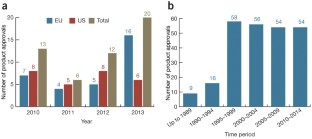
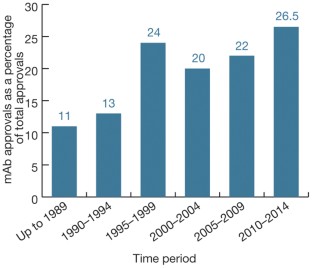
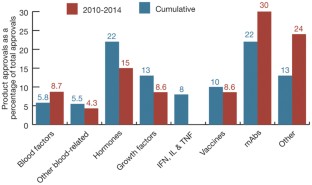
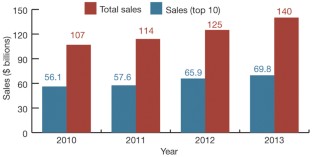
Monoclonal antibodies continue their march on the markets, optimized so-called biobetter versions of existing biologics are also gaining ground, but the rate of biosimilar approvals has seen a dramatic slowdown in recent years.
This is a preview of subscription content, access via your institution
Relevant articles
Open Access articles citing this article.
-
Protein overproduction alters exosome secretion in Chinese hamster ovary cells
Analytical and Bioanalytical Chemistry Open Access 09 May 2023
-
A cooperative nano-CRISPR scaffold potentiates immunotherapy via activation of tumour-intrinsic pyroptosis
Nature Communications Open Access 11 February 2023
-
Engineering Saccharomyces cerevisiae for the production and secretion of Affibody molecules
Microbial Cell Factories Open Access 09 March 2022
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
References
Walsh, G. Biopharmaceutical benchmarks. Nat. Biotechnol. 24, 769–776 (2006).
Walsh, G. Biopharmaceutical benchmarks 2010. Nat. Biotechnol. 28, 917–924 (2010).
Blackstone, E. & Fuhr, J. The Economics of biosimilars. American Health and Drug Benefits (July 2, 2014).
Rickwoood, S. & Di Biase, S. Searching for Terra Firma in the Biosimilars and Non-Original Biologics Market (IMS Health, Parsippany, NJ, USA, 2013).
Hospira Reports on success of its biosimilar proteins in Europe and Australia, La Meire Biologics 6, 3–4 (2013).
Dalgaard, K. et al. Biosimilars Seven Years On: Where are We and What's Next? (McKinsey and Company, New York, NY, USA, 2013).
Perez, H.L. et al. Antibody–drug conjugates: current status and future directions. Drug Discov. Today 19, 869–881 (2014).
Goede, V. et al. Comparison of Obinutuzumab (GA101) Plus Chlorambucil (Clb) Versus Rituximab Plus Clb in Patients with Chronic Lymphocytic Leukemia (CLL) and Co-Existing Medical Conditions (Comorbidities): Final Stage 2 Results of the CLL11 Trial. Presented at the 55th ASH Annual Meeting and Exposition. New Orleans, LA. December 7-10, 2013. Abstract 6.
Nielsen, L.S. et al. Single-batch production of recombinant human polyclonal antibodies. Mol. Biotechnol. 45, 257–266 (2010).
Kling, J. Sanofi to propel inhaled insulin Afrezza to market. Nat. Biotechnol. 32, 851–852 (2014).
Kwon, K.C. et al. Oral delivery of human biopharmaceuticals, autoantigens and vaccine antigens bioencapsulated in plant cells. Adv. Drug Deliv. Rev. 65, 782–799 (2013).
Emerton, D.A. Profitability in the biosimilars market: can you translate scientific excellence into a healthy commercial return? Bioprocess Int. 11, 6–14 (2013).
Brennan, T.A. & Wilson, J.M. The special case of gene therapy pricing. Nat. Biotechnol. 32, 874–876 (2014).
PhRMA. Medicines in Development: Biologics. 2013 report. http://www.phrma.org/sites/default/files/pdf/biologics2013.pdf (PhRMA, Washington, DC, 2013)
Olinger, G.G. et al. Delay treatment of Ebola virus infection with plant-derived monoclonal antibodies provides protection in rhesus monkeys. Proc. Natl. Acad. Sci. USA 109, 18030–18035 (2012).
Qui, X. et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 10.1038/nature13777 (29 August 2014).
Broz, A., Huang, N. & Unruh, G. Plant-based protein biomanufacturing. Genet. Eng. Biotechnol. News 33, 32–33 (2013).
Beck, A. & Reichert, J.M. Approval of the first biosimilar antibodies in Europe. MAbs 5, 621–623 (2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Supplementary Table 1
New traditional-type biotech product* approvals 2010-2014. (PDF 161 kb)
Supplementary Table 2
Stem cell products in phase 3 clinical trials (PDF 50 kb)
Rights and permissions
About this article
Cite this article
Walsh, G. Biopharmaceutical benchmarks 2014. Nat Biotechnol 32, 992–1000 (2014). https://doi.org/10.1038/nbt.3040
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3040
This article is cited by
-
A cooperative nano-CRISPR scaffold potentiates immunotherapy via activation of tumour-intrinsic pyroptosis
Nature Communications (2023)
-
Protein overproduction alters exosome secretion in Chinese hamster ovary cells
Analytical and Bioanalytical Chemistry (2023)
-
Knockout of caspase-7 gene improves the expression of recombinant protein in CHO cell line through the cell cycle arrest in G2/M phase
Biological Research (2022)
-
Engineering Saccharomyces cerevisiae for the production and secretion of Affibody molecules
Microbial Cell Factories (2022)
-
Mapping the molecular basis for growth related phenotypes in industrial producer CHO cell lines using differential proteomic analysis
BMC Biotechnology (2021)
