Abstract
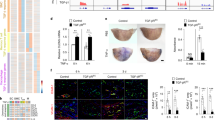
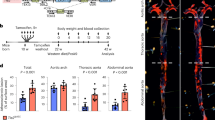
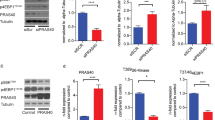
Recent studies suggest that statins can function to protect the vasculature in a manner that is independent of their lipid-lowering activity. We show here that statins rapidly activate the protein kinase Akt/PKB in endothelial cells. Accordingly, simvastatin enhanced phosphorylation of the endogenous Akt substrate endothelial nitric oxide synthase (eNOS), inhibited apoptosis and accelerated vascular structure formation in vitro in an Akt-dependent manner. Similar to vascular endothelial growth factor (VEGF) treatment, both simvastatin administration and enhanced Akt signaling in the endothelium promoted angiogenesis in ischemic limbs of normocholesterolemic rabbits. Therefore, activation of Akt represents a mechanism that can account for some of the beneficial side effects of statins, including the promotion of new blood vessel growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Goldstein, J.L. & Brown, M.S. Regulation of the mevalonate pathway. Nature 343, 425– 430 (1990).
ScandinavianSimvastatinSurvivalStudyGroup. Randomized trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 344, 1383–1389 (1994).
Levine, G.N., Keaney Jr., J.F. & Vita, J.A. Cholesterol reduction in cardiovascular disease. Clinical benefits and possible mechanisms. N. Engl. J. Med. 332, 512–521 ( 1995).
Sacks, F.M. et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N. Engl. J. Med. 335, 1001–1009 (1996).
WestofScotlandCoronaryPreventionStudyGroup. Influence of pravastatin and plasma lipids of clinical events in the West of Scotland Coronary Prevention Study (WOSCOPS). Circulation 97 , 1440–1445 (1998).
Endres, M. et al. Stroke protection by 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase inhibitors mediated by endothelial nitric oxide synthase. Proc. Natl. Acad. Sci. USA 95, 8880–8885 (1998).
Lefer, A.M. et al. Simvastatin preserves the ischemic-reperfused myocardium in normocholesterolemic rat hearts. Circulation 100, 178–184 (1999).
Pruefer, D., Scalia, R. & Lefer, A.M. Simvastatin inhibits leukocyte-endothelial cell interactions and protects against inflammatory processes in normocholesterolemic rats. Arterioscler. Thromb. Vasc. Biol. 19, 2894– 2900 (1999).
Osborne, J.A., Siegman, M.J., Sedar, A.W., Mooers, S.U. & Lefer, A.M. Lack of endothelium-dependent relaxation in coronary resistance arteries of cholesterol-fed rabbits. Am. J. Physiol. 256(3 Pt 1), C591– C597 (1989).
Lefer, A.M. & Lefer, D.J. The role of nitric oxide and cell adhesion molecules on the microcirculation in ischaemia-reperfusion. Cardiovasc. Res. 32, 743–751 (1996).
Dupuis, J., Tardif, J.-C., Cernacek, P. & Théroux, P. Cholesterol reduction rapidly improves endothelial function after acute coronary syndromes: the RECIFE (Reduction of Cholesterol in Ischemia and Function of the Endothelium) trial. Circulation 99, 3227–3233 (1999).
Baller, D. et al. Improvement in coronary flow reserve determined by positron emission tomography after 6 months of cholesterol-lowering therapy in patients with early stages of coronary atherosclerosis. Circulation 99, 2871–2875 (1999).
Datta, S.R., Brunet, A. & Greenberg, M.E. Cellular survival: a play in three Akts. Genes Dev. 13, 2905–2927 (1999).
Gerber, H.-P. et al. Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 3′-kinase/Akt signal transduction pathway: Requirement for Flk-1/KDR activation. J. Biol. Chem. 273, 30336–30343 (1998).
Fujio, Y. & Walsh, K. Akt mediates cytoprotection of endothelial cells by vascular endothelial growth factor in an anchorage-dependent manner . J. Biol. Chem. 274, 16349– 16354 (1999).
Kim, I. et al. Angiopoietin-1 regulates endothelial cell survival through the phosphatidylinositol 3′-kinase/Akt signal transduction pathway. Circ. Res. 86, 24–29 ( 2000).
Kontos, C.D. et al. Tyrosine 1101 of Tie2 is the major site of association of p85 and is required for activation of phosphatidylinositol 3-kinase and Akt . Mol. Cell. Biol. 18, 4131– 4140 (1998).
Papapetropoulos, A. et al. Angiopoietin-1 inhibits endothelial cell apoptosis via the Akt/Survivin pathway. J. Biol. Chem. 275, 9102–9105 (2000).
Carmeliet, P. et al. Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell 98, 147–157 ( 1999).
Fujio, Y., Nguyen, T., Wencker, D., Kitsis, R.N. & Walsh, K. Akt promotes survival of cardiomyocytes in vitro and protects against ischemia-reperfusion injury in mouse heart. Circulation 101, 660–667 (2000).
Fulton, D. et al. Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature 399, 597 –601 (1999).
Dimmeler, S. et al. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature 399, 601–605 (1999).
Luo, Z. et al. Acute modulation of endothelial Akt/PKB activity alters NO-dependent vasomotor activity in vivo. J. Clin. Invest., In press. (2000).
Morales-Ruiz, M. et al. Vascular endothelial growth factor-stimulated actin reorganization and migration of endothelial cells is regulated via the serine/threonine kinase Akt. Circ. Res. 86, 892– 896 (2000).
Benjamin, L.E., Golijanin, D., Itin, A., Pode, D. & Keshet, E. Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J. Clin. Invest. 103, 159–165 (1999).
Alon, T. et al. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity . Nature. Med. 1, 1024– 1028 (1995).
Ziche, M. et al. Nitric oxide synthase lies downstream from vascular endothelial growth factor-induced but not basic fibroblast growth factor-induced angiogenesis . J. Clin. Invest. 99, 2625– 2634 (1997).
Murohara, T. et al. Nitric oxide synthase modulates angiogenesis in response to tissue ischemia. J. Clin. Invest. 101, 2567 –2578 (1998).
Pu, L.Q. et al. Angiogenic growth factor and revascularization of the ischemic limb: evaluation in a rabbit model. J. Surg. Res. 54 , 575–583 (1993).
Simon, D.I. et al. Antiplatelet properties of protein S-nitrosothiols derived from nitric oxide and endothelium-derived relaxing factor. Arterioscler. Thromb. 13, 791–799 ( 1993).
Qian, H.S., Neplioueva, V., Shetty, G.A., Channon, K.M. & George, S.E. Nitric oxide synthase gene therapy rapidly reduces adhesion molecule expression and inflammatory cell infiltration in carotid arteries of cholesterol-fed rabbits. Circulation 99, 2979–2982 (1999).
von der Leyen, H.E. et al. Gene therapy inhibiting neointimal vascular lesion: in vivo transfer of endothelial cell nitric oxide synthase gene. Proc. Natl. Acad. Sci. USA 92, 1137– 1141 (1995).
Guo, K., Andrés, V. & Walsh, K. Nitric oxide-induced downregulation of cdk2 activity and cyclin A gene transcription in vascular smooth muscle cells. Circulation 20, 2066–2072 (1998).
Laufs, U. & Liao, J.K. Post-transcriptional regulation of endothelial nitric oxide synthase mRNA stability by Rho GTPase. J. Biol. Chem. 273, 24266–24271 (1998).
Kaesemeyer, W.H., Caldwell, R.B., Huang, J. & Caldwell, R.W. Pravastatin sodium activates endothelial nitric oxide synthase independent of its cholesterol-lowering actions. J. Amer. Coll. Cardiol. 33, 234–241 (1999).
TASCWorkingGroup. Investigation of patients with intermittent claudication. J. Vasc. Surg. 31 ( Suppl.), S62–S66 (2000).
Acknowledgements
This work was support by National Institutes of Health grants RO1-AR40197, RO1-HL50692, RO1-AG15052 and PO1-HD23681 to K.W. We thank Neil Tritman for supplying the human vascular cells.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kureishi, Y., Luo, Z., Shiojima, I. et al. The HMG-CoA reductase inhibitor simvastatin activates the protein kinase Akt and promotes angiogenesis in normocholesterolemic animals.. Nat Med 6, 1004–1010 (2000). https://doi.org/10.1038/79510
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/79510
This article is cited by
-
The protective effects of statins in traumatic brain injury
Pharmacological Reports (2024)
-
Paradoxical effects of statins on endothelial and cancer cells: the impact of concentrations
Cancer Cell International (2023)
-
Therapeutic angiogenesis and tissue revascularization in ischemic vascular disease
Journal of Biological Engineering (2023)
-
Association between atherogenic risk-modulating proteins and endothelium-dependent flow-mediated dilation in coronary artery disease patients
European Journal of Applied Physiology (2023)
-
Cumulative effect of simvastatin, l-arginine, and tetrahydrobiopterin on cerebral blood flow and cognitive function in Alzheimer’s disease
Alzheimer's Research & Therapy (2022)