Abstract
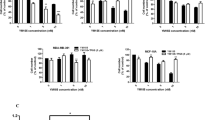
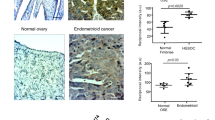
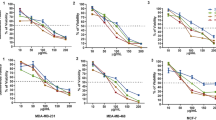
Superoxide dismutases (SOD) are essential enzymes that eliminate superoxide radical (O2-) and thus protect cells from damage induced by free radicals1,2,3. The active O2- production and low SOD activity in cancer cells3,4,5,6,7 may render the malignant cells highly dependent on SOD for survival and sensitive to inhibition of SOD. Here we report that certain oestrogen derivatives selectively kill human leukaemia cells but not normal lymphocytes. Using complementary DNA microarray and biochemical approaches, we identify SOD as a target of this drug action and show that chemical modifications at the 2-carbon (2-OH, 2-OCH3) of the derivatives are essential for SOD inhibition and for apoptosis induction. Inhibition of SOD causes accumulation of cellular O2- and leads to free-radical-mediated damage to mitochondrial membranes, the release of cytochrome c from mitochondria and apoptosis of the cancer cells. Our results indicate that targeting SOD may be a promising approach to the selective killing of cancer cells, and that mechanism-based combinations of SOD inhibitors with free-radical-producing agents may have clinical applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Halliwell, B. & Gutterridge, J. M. C. Free Radicals in Biology and Medicine (Oxford Univ. Press, New York, 1985).
Fridovich, I. Superoxide radical and superoxide dismutase. Annu. Rev. Biochem. 64, 97–112 ( 1995).
Oberley, L. W. & Buettner, G. R. Role of superoxide dismutase in cancer: a review. Cancer Res 39, 1141 –1149 (1979).
Dionisi, O., Galeotti, T., Terranova, T. & Azzi, A. Superoxide radicals and hydrogen peroxide formation in mitochondria from normal and neoplastic tissues. Biochim. Biophys. Acta 403, 292–300 (1975).
Marklund, S. L., Westman, N. G., Lundgren, E. & Roos, G. Copper- and zinc-containing superoxide dismutase, manganese-containing superoxide dismutase, catalase, and glutathione peroxidase in normal and neoplastic human cell lines and normal human tissues. Cancer Res. 42 , 1955–1961 (1982).
Van Driel, B. E. & Van Noorden, C. J. Oxygen insensitivity of the histochemical assay of glucose-6-phosphate dehydrogenase activity for the discrimination between nonmalignant and malignant cells. J. Histochem. Cytochem. 47, 575– 582 (1999).
Shulyakovskaya, T., Sumegi, L. & Gal, D. In vivo experimental studies on the role of free radicals in photodynamic therapy. I. Measurement of the steady state concentration of free radicals in tumor tissues of mice. Biochem. Biophys. Res. Commun. 95, 581–587 (1993).
Cushman, M., He, H. -M., Katzenellenbogen, J. A., Lin, C. M. & Hamel, E. Synthesis, antitubulin and antimitotic activity, and cytotoxicity of analogs of 2-methoxyestradiol, an endogenous mammalian metabolite of estradiol that inhibits tubulin polymerization by binding to the colchicine binding site. J. Med. Chem. 38, 2041–2049 (1995).
Mukhopadhyay, T. & Roth, J. A. Superinduction of wild-type p53 protein after 2-methoxyestradiol treatment of Ad5p53-transduced cells induces tumor cell apoptosis. Oncogene 17, 241–246 (1998).
Fujimura, M. et al. Manganese superoxide dismutase mediates the early release of mitochondrial cytochrome c and subsequent DNA fragmentation after permanent focal cerebral ischemia in mice. J. Neurosci. 19, 3414–3422 (1999).
Bindokas, V. J., Jordan, J., Lee, C. C. & Miller, R. J. Superoxide production in rat hippocampal neurons: selective imaging with hydroethidine. J. Neurosci. 16, 1324–1336 (1996).
Ukeda, H., Maeda, S., Ishii, T. & Sawamura, M. Spectrophotometric assay for superoxide dismutase based on tetrazolium salt 3′-1-[(phenylamino)-carbonyl]-3,4-tetrazolium-bis(4-methoxy-6-nitro) benzenesulfonic acid hydrate reduction by xanthine-xanthine oxidase. Anal. Biochem. 251, 206–209 (1997).
Zwacka, R. M., Dudus, L., Epperly, M. W., Greenberger, J. S. & Engelhardt, J. F. Redox gene therapy protects human IB-3 lung epithelial cells against ionizing radiation-induced apoptosis. Hum. Gene Ther. 9, 1381– 1386 (1998).
Heiden, M. G. V., Chandel, N. S., Williamson, E. K., Schumacker, P. T. & Thompson, C. B. Bcl-X L regulates the membrane potential and volume homeostasis of mitochondria. Cell 91, 627–637 (1997).
Marzo, I. et al. Bax and adenine nucleotide translocator cooperate in the mitochondrial control of apoptosis. Science 281, 2027– 2031 (1998).
Li, P. et al. Cytochrome c and ATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91, 479–489 (1997).
Rigo, A., Viglino, P. & Rotilio, G. Kinetic study of O2- dismutation by bovine superoxide dismutase: evidence for saturation of the catalytic sites by O2-. Biochem. Biophys. Res. Commun. 63, 1013–1018 ( 1975).
D'Amato, R. J., Lin, C. M., Flynn, E., Folkman, J. & Hamel, E. 2-methoxyestradiol, an endogenous mammalian metabolite, inhibits tubulin polymerization by interacting at the colchicine site. Proc. Natl Acad. Sci. USA 91, 3964– 3968 (1994).
Fotsis, T. et al. The endogenous oestrogen metabolite 2-methoxyoestradiol inhibits angiogenesis and supresses tumor growth. Nature 368 , 237–239 (1994).
Gutteridge, J. M., Rowley, D. A. & Halliwell, B. Superoxide-dependent formation of hydroxyl radicals and lipid peroxidation in the presence of iron salts. Detection of ‘catalytic’ iron and anti-oxidant activity in extracellular fluids. Biochem. J. 206, 605–609 ( 1982).
Hamana, K. et al. DNA strand scission by enzymatically reduced mitomycin C: evidence for participation of the hydroxyl radical in the DNA damage. Biochem. Int. 10, 301–309 (1985).
Pervaiz, S., Ramalingam, J. K., Hirpara, J. L. & Clement, M. Superoxide anion inhibits drug-induced tumor cell death. FEBS Lett. 459, 343–348 ( 1999).
Casppary, W. J., Niziak, C., Lanzo, D. A., Friedman, R. & Bachur, N. R. Bleomycin A2: a ferrous oxidase. Mol. Pharmacol. 16, 256–260 ( 1979).
Young, R. C., Ozols, R. F. & Myers, C. E. The anthracycline antineoplastic drugs. N. Engl. J. Med. 305, 139–153 (1981).
Gajewski, E., Rao, G., Nackerdien, Z. & Dizdaroglu, M. Modification of DNA bases in mammalian chromatin by radiation-generated free radicals. Biochemistry 29, 7876– 7882 (1990).
Mohanty, J. G., Jaffe, J. S., Schulman, E. S. & Raible, D. G. A highly sensitive fluorescent micro-assay of H2O2 release from activated human leukocytes using a dihydroxyphenoxazine derivative. J. Immunol. Methods 202, 133–141 (1997).
Huang, P., Chubb, S. & Plunkett, W. Termination of DNA synthesis by 9-β-D-arabinofuranosyl-2-fluoroadenine: a mechanism for cytotoxicity. J. Biol. Chem. 265, 16617–16625 (1990).
Acknowledgements
We thank J. Engelhardt and the Vector Core Facility of University of Iowa for Ad.CuZnSOD, Ad.MnSOD and the control viral vectors; M. Du and A. Sandoval for technical assistance in the isolation and preparation of primary cells from health donors and from leukaemia patients; and J. Richard for editorial assistance. This work was supported in part by NIH/NCI grants to P.H. and W.P.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, P., Feng, L., Oldham, E. et al. Superoxide dismutase as a target for the selective killing of cancer cells. Nature 407, 390–395 (2000). https://doi.org/10.1038/35030140
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35030140
This article is cited by
-
Purification, characterization, and antioxidant activity of polysaccharides from Grifola frondosa by hydrogen peroxide/ascorbic acid-assisted extraction
Journal of Food Measurement and Characterization (2024)
-
Electroactive membrane fusion-liposome for increased electron transfer to enhance radiodynamic therapy
Nature Nanotechnology (2023)
-
Expression Changes of SIRT1 and FOXO3a Significantly Correlate with Oxidative Stress Resistance Genes in AML Patients
Indian Journal of Hematology and Blood Transfusion (2023)
-
Pyrvinium pamoate inhibits cell proliferation through ROS-mediated AKT-dependent signaling pathway in colorectal cancer
Medical Oncology (2021)
-
Thymol induces mitochondrial pathway-mediated apoptosis via ROS generation, macromolecular damage and SOD diminution in A549 cells
Pharmacological Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.