Abstract
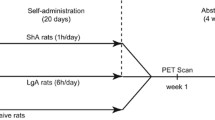
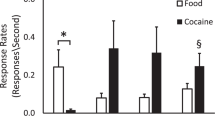
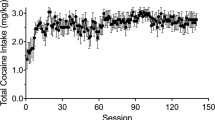
Cocaine addiction involves an escalation in drug intake which alters many brain functions. The present study documented cocaine-induced changes in brain metabolic activity as a function of cocaine self-administration history. Experimentally naive rhesus monkeys (N = 6) were given increasing access to cocaine under a fixed-ratio schedule of intravenous (i.v.) drug self-administration. PET imaging with F-18 labeled fluorodeoxyglucose (FDG) was used to measure acute intramuscular (i.m.) cocaine-induced changes in brain metabolism in the cocaine-naïve state, following 60 sessions under limited-access conditions (1 h/day), following 60 sessions under extended-access conditions (4 h/day), and following 4 weeks of drug withdrawal. In the cocaine-naïve state, cocaine-induced increases in brain metabolism were restricted to the prefrontal cortex. As cocaine exposure increased from limited to extended access, metabolic effects expanded throughout the frontal cortex and were induced within the striatum. Conversely, cocaine-induced activation was far less robust following withdrawal. The results highlight a progressive expansion of the metabolic effects of cocaine to include previously unaffected dopamine innervated brain regions as a consequence of cocaine self-administration history. The identification of brain regions progressively influenced by drug exposure may be highly relevant toward efforts to develop treatments for cocaine addiction.




Similar content being viewed by others
References
Breiter, H. C., Gollub, R. L., Weisskoff, R. M., Kennedy, D. N., Makris, N., Berke, J. D., et al. (1997). Acute effects of cocaine on human brain activity and emotion. Neuron, 19(3), 591–611.
Hayasaka, S., & Nichols, T. E. (2003). Validating cluster size inference: random field and permutation methods. Neuroimage, 20(4), 2343–2356.
Henry, P. K., Davis, M., & Howell, L. L. (2009). Effects of cocaine self-administration history under limited and extended access conditions on in vivo striatal dopamine neurochemistry and acoustic startle in rhesus monkeys. Psychopharmacology (Berl), 205(2), 237–247.
Howell, L. L., Hoffman, J. M., Votaw, J. R., Landrum, A. M., & Jordan, J. F. (2001). An apparatus and behavioral training protocol to conduct positron emission tomography (PET) neuroimaging in conscious rhesus monkeys. Journal of Neuroscience Methods, 106(2), 161–169.
Howell, L. L., Hoffman, J. M., Votaw, J. R., Landrum, A. M., Wilcox, K. M., & Lindsey, K. P. (2002). Cocaine-induced brain activation determined by positron emission tomography neuroimaging in conscious rhesus monkeys. Psychopharmacology (Berl), 159(2), 154–160.
Howell, L. L., Votaw, J. R., Goodman, M. M., & Lindsey, K. P. (2010). Cortical activation during cocaine use and extinction in rhesus monkeys. Psychopharmacology (Berl), 208(2), 191–199.
Kufahl, P. R., Li, Z., Risinger, R. C., Rainey, C. J., Wu, G., Bloom, A. S., et al. (2005). Neural responses to acute cocaine administration in the human brain detected by fMRI. Neuroimage, 28(4), 904–914.
London, E. D., Cascella, N. G., Wong, D. F., Phillips, R. L., Dannals, R. F., Links, J. M., et al. (1990). Cocaine-induced reduction of glucose utilization in human brain. A study using positron emission tomography and [fluorine 18]-fluorodeoxyglucose. Archives of General Psychiatry, 47(6), 567–574.
Lyons, D., Friedman, D. P., Nader, M. A., & Porrino, L. J. (1996). Cocaine alters cerebral metabolism within the ventral striatum and limbic cortex of monkeys. The Journal of Neuroscience, 16(3), 1230–1238.
Mathew, R. J., Wilson, W. H., Lowe, J. V., & Humphries, D. (1996). Acute changes in cranial blood flow after cocaine hydrochloride. Biological Psychiatry, 40, 609–616.
Porrino, L. J., Lyons, D., Miller, M. D., Smith, H. R., Friedman, D. P., Daunais, J. B., et al. (2002). Metabolic mapping of the effects of cocaine during the initial phases of self-administration in the nonhuman primate. The Journal of Neuroscience, 22(17), 7687–7694.
Porrino, L. J., Lyons, D., Smith, H. R., Daunais, J. B., & Nader, M. A. (2004). Cocaine self-administration produces a progressive involvement of limbic, association, and sensorimotor striatal domains. The Journal of Neuroscience, 24(14), 3554–3562.
Volkow, N. D., Fowler, J. S., Wolf, A. P., Hitzemann, R., Dewey, S., Bendriem, B., et al. (1991). Changes in brain glucose metabolism in cocaine dependence and withdrawal. The American Journal of Psychiatry, 148(5), 621–626.
Volkow, N. D., Hitzemann, R., Wang, G. J., Fowler, J. S., Wolf, A. P., Dewey, S. L., et al. (1992). Long-term frontal brain metabolic changes in cocaine abusers. Synapse, 11(3), 184–190.
Volkow, N. D., Wang, G. J., Fowler, J. S., Hitzemann, R., Angrist, B., Gatley, S. J., et al. (1999). Association of methylphenidate-induced craving with changes in right striato-orbitofrontal metabolism in cocaine abusers: implications in addiction. The American Journal of Psychiatry, 156(1), 19–26.
Volkow, N. D., Wang, G. J., Ma, Y., Fowler, J. S., Wong, C., Ding, Y. S., et al. (2005). Activation of orbital and medial prefrontal cortex by methylphenidate in cocaine-addicted subjects but not in controls: relevance to addiction. The Journal of Neuroscience, 25(15), 3932–3939.
Acknowledgements
Supported by USPHS grants DA016589, DA010344, DA00517, and RR00165.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Henry, P.K., Murnane, K.S., Votaw, J.R. et al. Acute brain metabolic effects of cocaine in rhesus monkeys with a history of cocaine use. Brain Imaging and Behavior 4, 212–219 (2010). https://doi.org/10.1007/s11682-010-9100-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-010-9100-5