Abstract
Background
Smoking continues to be a major contributor to the burden of disease across the world although there has been a decrease in some developed countries such as USA and Australia. In countries of South-East Asia with a high prevalence of smoking, the incidence of tobacco-related diseases will continue to increase.
Methods
We reviewed the literature in relation to the pharmacology of nicotine, the measures used to determine the efficacy of anti-smoking therapies, and the randomised controlled trials and systematic reviews of pharmacotherapies published between 2004 and 2010. We focused primarily on the three first line therapies that are currently available: nicotine replacement therapy (NRT), bupropion and varenicline.
Results
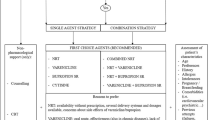
Randomised controlled trials and meta-analyses have demonstrated that single therapy with either NRT, bupropion or varenicline are all more effective than placebo for smoking cessation. Abstinence rates for monotherapies varies from 13.3% to 19% for NRT compared to 7.5% to 14% for placebo, 19% to 19.7% for bupropion versus 10.9% to 11% for placebo and 25.5% to 25.6% for varenicline versus 11.2% to 14.8% for placebo. Of current therapies varenicline appears to be more effective at achieving abstinence. Some combination therapies with one or two formulations of NRT or NRT plus bupropion have demonstrated superior results to monotherapy. To date there are no randomised controlled trials of varenicline in combination with NRT or bupropion.
Conclusion
Further studies are required to address the uncertainty that exists on the most appropriate duration of therapy as well as the effectiveness and safety of combination pharmacotherapy. Post-marketing surveillance continues to play an important role in monitoring the adverse effects events associated with these therapies.




Similar content being viewed by others
References
Winstanley M, White V. Chapter 1. Trends in the prevalence of smoking. In: Scollo MM, Winstanley MH, eds. Tobacco in Australia: Facts and Issues. 3 rd ed. Melbourne: Cancer Council Victoria; 2008. Accessed 22 December 2010 at http://www.tobaccoinaustralia.org.au/chapter-1-prevalence.
Centers for Disease Control and Prevention (CDC). Cigarette smoking among adults—United States, 2007. MMWR Morb Mortal Wkly Rep. 2008;57:1221–6.
WHO (World Health Organisation). Prevalence of current tobacco use among adults aged ≥ 15 years. Accessed 27 January 2010 at http://www.who.int/whosis/en/index.html
WHO (World Health Organisation). Report on the Global Tobacco Epidemic, 2009. Implementing smoke-free environments. Appendix VII: Age-Standardized Prevalence Estimates for Smoking, 2006. Accessed 27 January 2009 at www.who.int/tobacco/mpower/en
Gu D, Kelly TN, Wu X, Chen J, Samet JM, Huang J, et al. Mortality attributable to smoking in China. N Engl J Med. 2009;360:150–9.
Peto R, Lopez AD. The future worldwide health effects of current smoking patterns. In: Koop CE, Pearson CE, Schwarz MR, editors. Global health in the 21st century. New York: Jossey-Bass; 2000.
Frishman WH. Smoking cessation pharmacotherapy. Ther Adv Cardiovasc Dis. 2009;3:287–308.
SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tobacco Res. 2002;4:149–59.
Royal College of Physicians of London. Nicotine Addiction in Britain. A report of the Tobacco Advisory Group of the Royal College of Physicians, London. February 2000 (Accessed September 2009 at http://www.rcplondon.ac.uk/pubs/brochure.aspx?e=131)
Hubbard R, Lewis S, Smith C, Godfrey C, Smeeth L, Farrington P, et al. Use of nicotine replacement therapy and the risk of acute myocardial infarction, stroke, and death. Tobacco Control. 2005;14:416–21.
Stead LF, Perera R, Bullen C, Mant D, Lancaster T. Nicotine replacement therapy for smoking cessation. Cochrane Database of Systematic Reviews 2008, Issue 1. Art. No.: CD000146.
Silagy C, Lancaster T, Stead L, Mant D, Fowler G. Nicotine replacement therapy for smoking cessation. Cochrane Database Syst Rev 2004; 3, CD000146.
Schnoll RA et al. Effectiveness of extended-duration transdermal nicotine therapy: a randomized trial. Ann Intern Med. 2010;152:144.
Roddy E. ABC of smoking cessation. Bupropion and other non-nicotine pharmacotherapies. BMJ. 2004;328:509–11.
Hughes JR, Stead LF, Lancaster T. Antidepressants for smoking cessation. Cochrane Database of Systematic Reviews 2007, Issue 1. Art. No.: CD000031.
Hajek P, Stead LF, West R, Jarvis M, Lancaster T. Relapse prevention interventions for smoking cessation. Cochrane Database Syst Rev 2009; 1:CD003999.
FDA. The smoking cessation aids varenicline (marketed as Chantix) and bupropion (marketed as Zyban and generics). Drug Safety Newsletter. 2009;2:1–4.
Kuehn BM. Varenicline gets stronger warnings about psychiatric problems, vehicle crashes. JAMA. 2009;302:834.
Faessel HM, Smith BJ, Gibbs MA, Gobey JS, Clark DJ, Burstein AH. Single-dose pharmacokinetics of varenicline, a selective nicotinic receptor partial agonist, in healthy smokers and nonsmokers. J Clin Pharmacol. 2006;46:991–8.
Faessel HM, Gibbs MA, Clark DJ, Rohrbacher K, Stolar M, Burstein AH. Multiple-dose pharmacokinetics of the selective nicotinic receptor partial agonist varenicline in healthy smokers. J Clin Pharmacol. 2006;46:1439–48.
Burstein AH, Fullerton T, Clark DJ, Faessel HM. Pharmacokinetics, safety, and tolerability after single and multiple oral doses of varenicline in elderly smokers. J Clin Pharmacol. 2006;46:1234–40.
Obach RS, Reed-Hagen AE, Krueger SS, Obach BJ, O'Connell TN, Zandi KS, et al. Metabolism and disposition of varenicline, a selective alpha4beta2 acetylcholine receptor partial agonist, in vivo and in vitro. Drug Metab Dispos. 2006;34:121–30.
Garrison GD, Dugan SE. Varenicline: a first-line treatment option for smoking cessation. Clin Ther. 2009;31:463–91.
Cahill K, Stead LF, Lancaster T. Nicotine receptor partial agonists for smoking cessation. Cochrane Database of Systematic Reviews 2008, Issue 3. Art. No: CD006103.
Eisenberg MJ, Filion KB, Yavin D, Bélisle P, Mottillo S, Joseph L, et al. Pharmacotherapies for smoking cessation: a meta-analysis of randomized controlled trials. CMAJ. 2008;179:135–44.
Rigotti NA, Pipe AL, Benowitz NL, Arteaga C, Garza D, Tonstad S. Efficacy and safety of varenicline for smoking cessation in patients with cardiovascular disease: a randomized trial. Circulation. 2010;121:221–9.
Oncken C, Gonzales D, Nides M, Rennard S, Watsky E, Billing CB, et al. Efficacy and safety of the novel selective nicotinic acetylcholine receptor partial agonist, varenicline, for smoking cessation. Arch Intern Med. 2006;166:1571–7.
Harris DS, Anthenelli RM. Expanding treatment of tobacco dependence. Curr Psychiatry Rep. 2005;7:344–51.
Moore TJ, Cohen MR, Furberg CD. Strong Safety Signal Seen for New Varenicline Risks. Report. The Institute of Safe Medication Practices, Horsham, PA, USA. May 2008. Accessed 22 December 2009 at www.ismp.org/docs/vareniclineStudy.asp
Moore TJ, Furberg CD. Risk of psychiatric side effects with varenicline. BMJ. 2009;339:b4964.
Harrison-Woolrych M. Varenicline and suicide. Safety data from New Zealand. BMJ. 2009;339:b5654.
Stapleton JA, Watson L, Spirling LI, Smith R, Milbrandt A, Ratcliffe M, et al. Varenicline in the routine treatment of tobacco dependence: a pre-post comparison with nicotine replacement therapy and an evaluation in those with mental illness. Addiction. 2008;103:146–54.
Evins AE, Goff DC. Varenicline treatment for smokers with schizophrenia: a case series. J Clin Psychiatry. 2008;69:1016.
Purvis TL, Mambourg SE, Balvanz TM, Magallon HE, Pham RH. Safety and effectiveness of varenicline in a veteran population with a high prevalence of mental illness. Ann Pharmacother 2009; 43.
McClure JB, Swan GE, Jack L, Catz SL, Zbikowski SM, McAfee TA, et al. Mood side-effects and smoking outcomes among persons with or without probable lifetime depression taking varenicline. J Gen Intern Med. 2009;24:563–9.
Gunnell D, Irvine D, Wise L, Davies C, Martin RM. Varenicline and suicidal behaviour: a cohort study based on data from the general practice research database. BMJ. 2009;339:b3805.
Cornuz J, Zwahlen S, Jungi WF, Osterwalder J, Klingler K, van Melle G, et al. A vaccine against nicotine for smoking cessation: a randomized controlled trial. PLoS. 2008;3:e2547.
Shah SD, Wilken LA, Winkler SR, Lin SJ. Systematic review and meta-analysis of combination therapy for smoking cessation. J Am Pharm Assoc. 2008;48:659–65.
Piper ME, Smith SS, Schlam TR, Fiore MC, Jorenby DE, Fraser D, et al. Randomized placebo-controlled clinical trial of 5 smoking cessation pharmacotherapies. Arch Gen Psychiatry. 2009;66:1253–62.
Steinberg M, Greenhaus S, Schmelzer AC, Bover MT, Foulds J, Hoover DR, et al. Triple-combination pharmacotherapy for medically Ill smokers. A randomized trial. Ann Intern Med. 2009;150:447–54.
Smith SS, McCarthy DE, Japuntich SJ, Christiansen B, Piper ME, Jorenby DE, et al. Comparative effectiveness of 5 smoking cessation pharmacotherapies in primary care clinics. Arch Intern Med. 2009;169:2148–55.
Woolacott NF, Jones L, Forbes CA, Mather LC, Sowden AJ, Song FJ et al. The clinical effectiveness and cost-effectiveness of bupropion and nicotine replacement therapy for smoking cessation: a systematic review and economic evaluation. Health Technology Assessment 2002; 6(16). (Assessed November 2009 at http://www.hta.nhs.uk/pdfexecs/summ616.pdf )
Keating GM, Lyseng-Williamson KA. Varenicline: a pharmacoeconomic review of its use as an aid to smoking cessation. PharmacoEconomics. 2010;28:231–54.
Hammond EC, Horn D. Smoking and death rates. Report on forty-four months of follow up of 187,783 men. JAMA. 1958;166:1159–72–1294–308.
Quaak M, van Schayck CP, Knaapen AM, van Schooten FJ. Genetic variation as a predictor of smoking cessation success. A promising preventive and intervention tool for chronic respiratory diseases? Eur Respir J. 2009;33:468–80.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McNeil, J.J., Piccenna, L. & Ioannides-Demos, L.L. Smoking Cessation–Recent Advances. Cardiovasc Drugs Ther 24, 359–367 (2010). https://doi.org/10.1007/s10557-010-6246-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-010-6246-8