Abstract
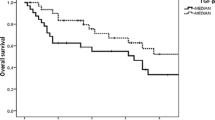
Introduction Studies investigating the prognostic effect of circulating TGF-β-1 in breast cancer have given inconsistent findings. The purpose of this study is to evaluate whether circulating transforming growth factor beta 1 (TGF-β-1) is associated with overall and disease-free survival in a cohort of recently diagnosed breast cancer patients. Methods We measured TGF-β-1 levels in plasma samples of breast cancer patients in the Shanghai Breast Cancer Study, a population-based case–control study. We evaluated the relationship between TGF-β-1 levels and overall and disease-free survival. The median follow up time was 7.2 years. Results We observed that, compared with the patients with the lowest quartile of plasma TGF-β-1, patients with the highest quartile of plasma TGF-β-1 had significantly worse overall survival with hazards ratio (HR) = 2.78, with 95% confidence interval (CI): 1.34–5.79 and disease-free survival with HR = 2.49, 95% CI: 1.15–5.41, while the patients with the second and third quartiles of plasma TGF-β-1 did not have significantly different overall and disease-free breast cancer survival. The shape of association between plasma TGF-β-1 levels and breast cancer survival appears to be non-linear. Stratified analysis by stage of disease did not appreciably change the association pattern. Conclusions We conclude that the relationship between circulating levels of TGF-β-1 and prognosis in breast cancer is complex and non-linear. High levels of TGF-β-1 are associated with worse survival independent of stage of disease.


Similar content being viewed by others
References
Massague J, Chen YG (2000) Controlling TGF-beta signaling. Genes Dev 14:627–644
Massague J (1996) TGFbeta signaling: receptors, transducers, and Mad proteins. Cell 85:947–950
Grau AM, Zhang L, Wang W et al (1997) Induction of p21waf1 expression and growth inhibition by transforming growth factor beta involve the tumor suppressor gene DPC4 in human pancreatic adenocarcinoma cells. Cancer Res 57:3929–3934
Hahn SA, Schutte M, Hoque AT et al (1996) DPC4, a candidate tumor suppressor gene at human chromosome 18q21.1. Science 271:350–353
Hunt KK, Fleming JB, Abramian A et al (1998) Overexpression of the tumor suppressor gene Smad4/DPC4 induces p21waf1 expression and growth inhibition in human carcinoma cells. Cancer Res 58:5656–5661
Bakin AV, Tomlinson AK, Bhowmick NA et al (2000) Phosphatidylinositol 3-kinase function is required for transforming growth factor beta-mediated epithelial to mesenchymal transition and cell migration. J Biol Chem 275:36803–36810
Teraoka H, Sawada T, Nishihara T et al (2001) Enhanced VEGF production and decreased immunogenicity induced by TGF-beta 1 promote liver metastasis of pancreatic cancer. Br J Cancer 85:612–617
Arteaga CL (2006) Inhibition of TGFbeta signaling in cancer therapy. Curr Opin Genet Dev 16:30–37
Bierie B, Moses HL (2006) Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nature Rev 6:506–520
Stuelten CH, DaCosta Byfield S, Arany PR et al (2005) Breast cancer cells induce stromal fibroblasts to express MMP-9 via secretion of TNF-alpha and TGF-beta. J Cell Sci 118:2143–2153
Kao JY, Gong Y, Chen CM et al (2003) Tumor-derived TGF-beta reduces the efficacy of dendritic cell/tumor fusion vaccine. J Immunol 170:3806–3811
Wakefield LM, Letterio JJ, Chen T et al (1995) Transforming growth factor-beta1 circulates in normal human plasma and is unchanged in advanced metastatic breast cancer. Clin Cancer Res 1:129–136
Tsushima H, Ito N, Tamura S et al (2001) Circulating transforming growth factor beta 1 as a predictor of liver metastasis after resection in colorectal cancer. Clin Cancer Res 7:1258–1262
Ivanovic V, Todorovic-Rakovic N, Demajo M et al (2003) Elevated plasma levels of transforming growth factor-beta 1 (TGF-beta 1) in patients with advanced breast cancer: association with disease progression. Eur J Cancer 39:454–461
Decensi A, Torrisi R, Fontana V et al (1998) Correlation between plasma transforming growth factor-beta 1 and second primary breast cancer in a chemoprevention trial. Eur J Cancer 34:999–1003
Shariat SF, Shalev M, Menesses-Diaz A et al (2001) Preoperative plasma levels of transforming growth factor beta(1) (TGF-beta(1)) strongly predict progression in patients undergoing radical prostatectomy. J Clin Oncol 19:2856–2864
Okumoto K, Hattori E, Tamura K et al (2004) Possible contribution of circulating transforming growth factor-beta1 to immunity and prognosis in unresectable hepatocellular carcinoma. Liver Int 24:21–28
Kong FM, Anscher MS, Murase T et al (1995) Elevated plasma transforming growth factor-beta 1 levels in breast cancer patients decrease after surgical removal of the tumor. Ann Surg 222:155–162
Sheen-Chen SM, Chen HS, Sheen CW et al (2001) Serum levels of transforming growth factor beta1 in patients with breast cancer. Arch Surg 136:937–940
Lebrecht A, Grimm C, Euller G et al (2004) Transforming growth factor beta 1 serum levels in patients with preinvasive and invasive lesions of the breast. Int J Biol Markers 19:236–239
Sminia P, Barten AD, van Waarde MA et al (1998) Plasma transforming growth factor beta levels in breast cancer patients. Oncol Rep 5:485–488
Ivanovic V, Demajo M, Krtolica K et al (2006) Elevated plasma TGF-beta1 levels correlate with decreased survival of metastatic breast cancer patients. Clin Chim Acta 371:191–193
Gao YT, Shu XO, Dai Q et al (2000) Association of menstrual and reproductive factors with breast cancer risk: results from the Shanghai Breast Cancer Study. Int J Cancer 87:295–300
Biswas S, Guix M, Rinehart C et al (2007) Inhibition of TGF-beta with neutralizing antibodies prevents radiation-induced acceleration of metastatic cancer progression. J Clin Invest
Harrell FEJ (2001) Regression modeling strategies, with applications to linear models, logistic regression, and survival analysis. Springer, New York
Hayes DF, Ethier S, Lippman ME (2006) New guidelines for reporting of tumor marker studies in breast cancer research and treatment: REMARK. Breast Cancer Res Treat 100:237–238
Lu H, Shu XO, Cui Y et al (2005) Association of genetic polymorphisms in the VEGF gene with breast cancer survival. Cancer Res 65:5015–5019
Parrinello G, Licata A, Colomba D et al (2005) Left ventricular filling abnormalities and obesity-associated hypertension: relationship with overproduction of circulating transforming growth factor beta1. J Hum Hypertens19:543–550
Zheng W, Chow WH, Yang G et al (2005) The Shanghai Women’s Health Study: rationale, study design, and baseline characteristics. Am J Epidemiol 162:1123–1131
Li C, Guo B, Wilson PB et al (2000) Plasma levels of soluble CD105 correlate with metastasis in patients with breast cancer. Int J Cancer 89:122–126
Nikolic-Vukosavljevic D, Todorovic-Rakovic N, Demajo M et al (2004) Plasma TGF-beta1-related survival of postmenopausal metastatic breast cancer patients. Clin Exp Metastasis 21:581–585
Jeon JH, Kim YS, Choi EJ et al (2001) Implication of co-measured platelet factor 4 in the reliability of the results of the plasma transforming growth factor-beta 1 measurement. Cytokine 16:102–105
Desruisseau S, Palmari J, Giusti C et al (2006) Determination of TGFbeta1 protein level in human primary breast cancers and its relationship with survival. Br J Cancer 94:239–246
Grainger DJ, Heathcote K, Chiano M et al (1999) Genetic control of the circulating concentration of transforming growth factor type beta1. Hum Mol Genet 8:93–97
Yokota M, Ichihara S, Lin TL et al (2000) Association of a T29–>C polymorphism of the transforming growth factor-beta1 gene with genetic susceptibility to myocardial infarction in Japanese. Circulation 101:2783–2787
Shu XO, Gao YT, Cai Q et al (2004) Genetic polymorphisms in the TGF-beta 1 gene and breast cancer survival: a report from the Shanghai Breast Cancer Study. Cancer Res 64:836–839
Yang YA, Dukhanina O, Tang B et al (2002) Lifetime exposure to a soluble TGF-beta antagonist protects mice against metastasis without adverse side effects. J Clin Invest 109:1607–1615
Acknowledgments
Research was supported in part by grant P20RR011792 from the NIH and RCMI, and by RO1 CA64277 from the NCI. We thank Drs. Fan Jin and Qi Dai for valuable contribution in coordinating the filed operation and Regina Courtney and Qing Wang for excellent technical supports. We are grateful to the patients and research staff who participated in the Shanghai Breast Cancer Study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grau, A.M., Wen, W., Ramroopsingh, D.S. et al. Circulating transforming growth factor-β-1 and breast cancer prognosis: results from the Shanghai Breast Cancer Study. Breast Cancer Res Treat 112, 335–341 (2008). https://doi.org/10.1007/s10549-007-9845-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9845-8