Abstract
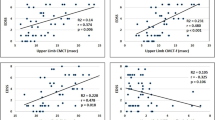
We designed this study to investigate possible correlations between variables measuring primary motor cortex excitability detected by single and paired-pulse transcranial magnetic stimulation (TMS) and the severity of clinical manifestations in patients with multiple sclerosis (MS). Thirty patients with MS in remission, 16 with relapsing–remitting (RR), 14 with secondary progressive disease (SP) and 17 healthy subjects participated in the study. In each subject, the central motor conduction time (CMCT) was calculated, and single-pulse and paired-pulse TMS at 3 and 10 ms interstimulus intervals was delivered over the primary motor cortex of the dominant hemisphere to measure the amplitude of motor-evoked potentials (MEPs), motor threshold (MTh), intracortical inhibition (ICI) and facilitation (ICF). Correlations were determined between the patients’ TMS findings and magnetic resonance imaging (MRI) (lesion load) and clinical features (expanded disability status scale, EDSS score). EDSS scores were significantly higher in SPMS than in RRMS patients. The MTh was significantly higher, and the MEP was significantly smaller in SPMS patients than in RRMS patients and control subjects. All patients had longer CMCTs than healthy subjects. In all patients, paired-pulse TMS elicited an inhibited test MEP at the 3-ms ISI and a facilitated test MEP at the 10 ms ISI. Post hoc analysis showed that ICI was significantly lower in SPMS patients than in those with RRMS and healthy subjects. EDSS scores correlated significantly with TMS measures (MEP, ICI, CMCT and MTh), but not with MRI lesion load. It was found that intracortical excitability as measured with TMS differs according to the clinical course of MS; it remains normal in patients with low EDSS scores and is altered in patients with high EDSS scores.

Similar content being viewed by others
References
Berardelli A, Inghilleri M, Cruccu G, Fornarelli M, Accornero N, Manfredi M (1988) Stimulation of motor tract in multiple sclerosis. J Neurol Neurosurg Psychiatry 51:677–683
Boniface SJ (2001) Plasticity after acute ischaemic stroke studied by transcranial magnetic stimulation. J Neurol Neurosurg Psychiatry 71:713–715
Calabrese M, Atzori M, Bernardi V et al (2007) Cortical atrophy is relevant in multiple sclerosis at clinical onset. J Neurol 254:1212–1220
Ciccarelli O, Brex PA, Thompson AJ, Miller DH (2002) Disability and lesion load in MS: a reassessment with MS functional composite score and 3D fast FLAIR. J Neurol 249:18–24
Caramia MD, Palmieri MG, Desiato MT et al (2004) Brain excitability changes in the relapsing and remitting phases of multiple sclerosis: a study with transcranial magnetic stimulation. Clin Neurophysiol 15:956–965
Classen J, Schnitzler A, Binkofski F et al (1997) The motor syndrome associated with exaggerated inhibition within the primary motor cortex of patients with hemiparetic stroke. Brain 120:605–619
Curra A, Modugno N, Inghilleri M, Manfredi M, Hallett M, Berardelli A (2002) Transcranial magnetic stimulation techniques in clinical investigation. Neurology 59:1851–1859
Dehmeshki J, Chard DT, Leary SM et al (2003) The normal appearing grey matter in primary progressive multiple sclerosis: a magnetisation transfer imaging study. J Neurol 250:67–74
Facchetti D, Mai R, Micheli A et al (1997) Motor evoked potentials and disability in secondary progressive multiple sclerosis. Can J Neurol Sci 24:332–337
Fierro B, Salemi G, Brighina F et al (2002) A transcranial magnetic stimulation study evaluating methylprednisolone treatment in multiple sclerosis. Acta Neurol Scand 105:152–157
Fuhr P, Kappos L (2001) Evoked potentials for evaluation of multiple sclerosis. Clin Neurophysiol 112:2185–2189
Geurts GJ, Barkhof F (2008) Grey matter pathology in multiple sclerosis. Lancet Neurol 7:841–851
Grimaud J, Lai M, Thorpe J et al (1996) Quantification of MRI lesion load in multiple sclerosis: a comparison of three computer-assisted techniques. Magn Reson Imaging 14:495–505
Hobart J, Kalkers N, Barkhof F, Uitdehaag B, Polman C, Thompson A (2004) Outcome measures for multiple sclerosis clinical trials: relative measurement precision of the expanded disability status scale and multiple sclerosis functional composite. Mult Scler 10:41–46
Humm AM, Truffert A, Magistris MR, Rösler KM (2002) Central motor conduction in acute relapsing–remitting versus chronic progressive multiple sclerosis. J Neurol 249:I/101–I/102
Humm AM, Magistris MR, Truffert A, Hess CW, Rösler KM (2003) Central motor conduction differs between acute relapsing–remitting and chronic progressive multiple sclerosis. Clin Neurophysiol 114:2196–2203
Humm AM, Z’Graggen WJ, Bühler R, Magistris MR, Rösler KM (2006) Quantification of central motor conduction deficits in multiple sclerosis patients before and after treatment of acute exacerbation by methylprednisolone. J Neurol Neurosurg Psychiatry 77:345–350
Kandler RH, Jarratt JA, Davies-Jones GA et al (1991) The role of magnetic stimulation as a quantifier of motor disability in patients with multiple sclerosis. J Neurol Sci 106:31–34
Khaleeli Z, Cercignani M, Audoin B et al (2007) Localized grey matter damage in early primary progressive multiple sclerosis contributes to disability. Neuroimage 37:253–261
Kidd D, Thompson PD, Day BL et al (1998) Central motor conduction time in progressive multiple sclerosis. Correlations with MRI and disease activity. Brain 121:1109–1116
Kujirai T, Caramia MD, Rothwell JC et al (1993) Corticocortical inhibition in human motor cortex. J Physiol 471:501–519
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Liepert J, Mingers D, Heesen C, Baumer T, Weiller C (2005) Motor cortex excitability and fatigue in multiple sclerosis: a transcranial magnetic stimulation study. Mult Scler 11:316–321
Lublin FD, Reingold SC (1996) Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on clinical trials of new agents in multiple sclerosis. Neurology 46:907–911
Mainero C, Inghilleri M, Pantano P et al (2004) Enhanced brain motor activity in patients with MS after a single dose of 3,4-diaminopyridine. Neurology 62:2044–2050
Masek M, Vaneckova M, Krasensky J et al (2008) Secondary-progressive form of multiple sclerosis: MRI changes versus clinical status. Neuro Endocrinol Lett 29:461–466
Oreja-Guevara C, Charil A, Caputo D et al (2006) Magnetization transfer magnetic resonance imaging and clinical changes in patients with relapsing-remitting multiple sclerosis. Arch Neurol 63:736–740
Rovaris M, Judica E, Gallo A et al (2006) Grey matter damage predicts the evolution of primary progressive multiple sclerosis at 5 years. Brain 129:2628–2634
Schmierer K, Irlbacher K, Grosse P, Röricht S, Meyer BU (2002) Correlates of disability in multiple sclerosis detected by transcranial magnetic stimulation. Neurology 59:1218–1224
Sheean GL, Murray NMF, Rothwell JC, Miller DH, Thompson AJ (1997) An electrophysiological study of the mechanism of fatigue in multiple sclerosis. Brain 120:299–315
Talairach J, Tournoux P (1988) Coplanar stereotaxic atlas of the human brain. Thieme, Stuttgart
Tassinari CA, Cincotta M, Zaccara G, Michelacci R (2003) Transcranial magnetic stimulation and epilepsy. Clin Neurophysiol 114:777–798
Vrenken H, Pouwels PJ, Geurts JJ et al (2006) Altered diffusion tensor in multiple sclerosis normal-appearing brain tissue: cortical diffusion changes seem related to clinical deterioration. J Magn Reson Imaging 23:628–636
Conflict of interest statement
The authors have reported no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Conte, A., Lenzi, D., Frasca, V. et al. Intracortical excitability in patients with relapsing–remitting and secondary progressive multiple sclerosis. J Neurol 256, 933–938 (2009). https://doi.org/10.1007/s00415-009-5047-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-009-5047-0