Abstract
Rationale
While cocaine blocks dopamine and serotonin transporters, considerably less emphasis has been placed on its effects following blockade of the norepinephrine transporter (NET). To date, no studies have made a systematic investigation of the effects of chronic cocaine on primate NET density.
Objective
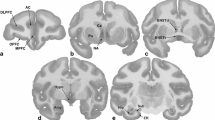
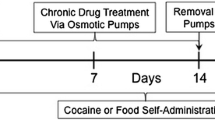
We previously reported increases in NET density in portions of the monkey bed nucleus of stria terminalis after 100 days of cocaine self-administration. In the present study we extend these findings and assess the changes in [3H]nisoxetine binding in additional brain regions of rhesus monkeys chronically self-administrating cocaine.
Results
[3H]Nisoxetine binding sites in the A1 noradrenergic cell group were significantly higher after 5 days of cocaine exposure. One hundred days of self-administration also induced a higher density of NET binding within the A1 cell group; however, in addition, the effects extended to the nucleus prepositus, as well as forebrain regions such as hypothalamic nuclei, basolateral amygdala, parasubiculum, and entorhinal cortex.
Conclusions
These data demonstrate that cocaine self-administration alters the noradrenergic system of nonhuman primates. Although cocaine affected NET binding sites in the forebrain projections of both the ventral (VNAB) and dorsal (DNAB) noradrenergic bundles, the alteration in the A1 cell group at the early time-point suggests that the VNAB appears to be more sensitive than the DNAB to the effects of cocaine. Given the role of norepinephrine in arousal and attention, as well as mediating responses to stress, long-term exposure to cocaine is likely to result in significant changes in the way in which information is perceived and processed by the central nervous system of long-term cocaine users.


Similar content being viewed by others
References
Alheid GF, Heimer L (1988) New perspectives in basal forebrain organization of special relevance for neuropsychiatric disorders: the striatopallidal, amygdaloid, and corticopetal components of substantia innominata. Neuroscience 27:1–39
Alheid GF, Beltramino CA, De Olmos JS, Forbes MS, Swanson DJ, Heimer L (1998) The neuronal organization of the supracapsular part of the stria terminalis in the rat: the dorsal component of the extended amygdala. Neuroscience 84:967–996
Arroyo M, Baker WA, Everitt BJ (2000) Cocaine self-administration in rats differentially alters mRNA levels of the monoamine transporters and striatal neuropeptides. Brain Res Mol Brain Res 83:107–120
Aston-Jones G, Delfs JM, Druhan J, Zhu Y (1999) The bed nucleus of the stria terminalis. A target site for noradrenergic actions in opiate withdrawal. Ann N Y Acad Sci 877:486–498
Belej T, Manji D, Sioutis S, Barros HM, Nobrega JN (1996) Changes in serotonin and norepinephrine uptake sites after chronic cocaine: pre- vs. post-withdrawal effects. Brain Res 736:287–296
Benmansour S, Tejani-Butt SM, Hauptmann M, Brunswick DJ (1992) Lack of effect of high-dose cocaine on monoamine uptake sites in rat brain measured by quantitative autoradiography. Psychopharmacology (Berl) 106:459–462
Bennett BA, Wichems CH, Hollingsworth CK, Davies HM, Thornley C, Sexton T, Childers SR (1995) Novel 2-substituted cocaine analogs: uptake and ligand binding studies at dopamine, serotonin and norepinephrine transport sites in the rat brain. J Pharmacol Exp Ther 272:1176–1186
Beveridge TJR, Smith HR, Nader MA, Porrino LJ (2004) Functional effects of cocaine self-administration in primate brain regions regulating cardiovascular function. Neurosci Lett 370:201–205
Broadbear JH, Winger G, Woods JH (1999) Cocaine-reinforced responding in rhesus monkeys: pharmacological attenuation of the hypothalamic–pituitary–adrenal axis response. J Pharmacol Exp Ther 290:1347–1355
Burchett SA, Bannon MJ (1997) Serotonin, dopamine and norepinephrine transporter mRNAs: heterogeneity of distribution and response to ‘binge’ cocaine administration. Brain Res Mol Brain Res 49:95–102
Calogero AE, Gallucci WT, Kling MA, Chrousos GP, Gold PW (1989) Cocaine stimulates rat hypothalamic corticotropin-releasing hormone secretion in vitro. Brain Res 505:7–11
Delfs JM, Zhu Y, Druhan JP, Aston-Jones G (2000) Noradrenaline in the ventral forebrain is critical for opiate withdrawal-induced aversion. Nature 403:430–434
de Wit H, Wise RA (1977) Blockade of cocaine reinforcement in rats with the dopamine receptor blocker pimozide, but not with the noradrenergic blockers phentolamine or phenoxybenzamine. Can J Psychol 31:195–203
Erb S, Stewart J (1999) A role for the bed nucleus of the stria terminalis, but not the amygdala, in the effects of corticotropin-releasing factor on stress-induced reinstatement of cocaine seeking. J Neurosci 19:RC35
Erb S, Hitchcott PK, Rajabi H, Mueller D, Shaham Y, Stewart J (2000) Alpha-2 adrenergic receptor agonists block stress-induced reinstatement of cocaine seeking. Neuropsychopharmacology 23:138–150
Erb S, Salmaso N, Rodaros D, Stewart J (2001) A role for the CRF-containing pathway from central nucleus of the amygdala to bed nucleus of the stria terminalis in the stress-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 158:360–365
Ferry B, Roozendaal B, McGaugh JL (1999) Basolateral amygdala noradrenergic influences on memory storage are mediated by an interaction between beta- and alpha1-adrenoceptors. J Neurosci 19:5119–5123
Forman LJ, Estilow S (1988) Cocaine influences beta-endorphin levels and release. Life Sci 43:309–315
Giros B, Jaber M, Jones SR, Wightman RM, Caron MG (1996) Hyperlocomotion and indifference to cocaine and amphetamine in mice lacking the dopamine transporter. Nature 379:606–612
Goeders NE (2002) The HPA axis and cocaine reinforcement. Psychoneuroendocrinology 27:13–33
Harris GC, Williams JT (1992) Sensitization of locus ceruleus neurons during withdrawal from chronic stimulants and antidepressants. J Pharmacol Exp Ther 261:476–483
Kampman KM, Volpicelli JR, Mulvaney F, Alterman AI, Cornish J, Gariti P, Cnaan A, Poole S, Muller E, Acosta T, Luce D, O’Brien C (2001) Effectiveness of propranolol for cocaine dependence treatment may depend on cocaine withdrawal symptom severity. Drug Alcohol Depend 63:69–78
Karoum F, Suddath RL, Wyatt RJ (1990) Chronic cocaine and rat brain catecholamines: long-term reduction in hypothalamic and frontal cortex dopamine metabolism. Eur J Pharmacol 186:1–8
Kleven MS, Koek W (1997) Discriminative stimulus properties of cocaine: enhancement by beta-adrenergic receptor antagonists. Psychopharmacology (Berl) 131:307–312
Kleven MS, Koek W (1998) Discriminative stimulus properties of cocaine: enhancement by monoamine reuptake blockers. J Pharmacol Exp Ther 284:1015–1025
Lee B, Tiefenbacher S, Platt DM, Spealman RD (2004) Pharmacological blockade of alpha2-adrenoceptors induces reinstatement of cocaine-seeking behavior in squirrel monkeys. Neuropsychopharmacology 29:686–693
Leri F, Flores J, Rodaros D, Stewart J (2002) Blockade of stress-induced but not cocaine-induced reinstatement by infusion of noradrenergic antagonists into the bed nucleus of the stria terminalis or the central nucleus of the amygdala. J Neurosci 22:5713–5718
Letchworth SR, Nader MA, Smith HR, Friedman DP, Porrino LJ (2001) Progression of changes in dopamine transporter binding site density as a result of cocaine self-administration in rhesus monkeys. J Neurosci 21:2799–2807
Levy AD, Li QA, Kerr JE, Rittenhouse PA, Milonas G, Cabrera TM, Battaglia G, Alvarez Sanz MC, Van de Kar LD (1991) Cocaine-induced elevation of plasma adrenocorticotropin hormone and corticosterone is mediated by serotonergic neurons. J Pharmacol Exp Ther 259:495–500
Little KY, Kirkman JA, Carroll FI, Clark TB, Duncan GE (1993) Cocaine use increases [3H]WIN 35428 binding sites in human striatum. Brain Res 628:17–25
Macey DJ, Smith HR, Nader MA, Porrino LJ (2003) Chronic cocaine self-administration upregulates the norepinephrine transporter and alters functional activity in the bed nucleus of the stria terminalis of the rhesus monkey. J Neurosci 23:12–16
Martin LJ, Powers RE, Dellovade TL, Price DL (1991) The bed nucleus-amygdala continuum in human and monkey. J Comp Neurol 309:445–485
Mash DC, Pablo J, Ouyang Q, Hearn WL, Izenwasser S (2002) Dopamine transport function is elevated in cocaine users. J Neurochem 81:292–300
McDougle CJ, Black JE, Malison RT, Zimmermann RC, Kosten TR, Heninger GR, Price LH (1994) Noradrenergic dysregulation during discontinuation of cocaine use in addicts. Arch Gen Psychiatry 51:713–719
Nader MA, Daunais JB, Moore T, Nader SH, Moore RJ, Smith HR, Friedman DP, Porrino LJ (2002) Effects of cocaine self-administration on striatal dopamine systems in rhesus monkeys: initial and chronic exposure. Neuropsychopharmacology 27:35–46
Paxinos G, Huang XF, Toga AW (2000) The rhesus monkey brain in stereotaxic coordinates. Academic Press, San Diego
Porrino LJ, Lyons D, Miller MD, Smith HR, Friedman DP, Daunais JB, Nader MA (2002) Metabolic mapping of the effects of cocaine during the initial phases of self-administration in the nonhuman primate. J Neurosci 22:7687–7694
Porrino LJ, Daunais JB, Smith HR, Nader MA (2004) The expanding effects of cocaine: studies in a nonhuman primate model of cocaine self-administration. Neurosci Biobehav Rev 27:813–820
Ritz MC, Cone EJ, Kuhar MJ (1990) Cocaine inhibition of ligand binding at dopamine, norepinephrine and serotonin transporters: a structure–activity study. Life Sci 46:635–645
Rivier C, Lee S (1994) Stimulatory effect of cocaine on ACTH secretion: role of the hypothalamus. Mol Cell Neurosci 5:189–195
Roder S, Ciriello J (1993) Innervation of the amygdaloid complex by catecholaminergic cell groups of the ventrolateral medulla. J Comp Neurol 332:105–122
Roder S, Ciriello J (1994) Collateral axonal projections to limbic structures from ventrolateral medullary A1 noradrenergic neurons. Brain Res 638:182–188
Rogers RD, Robbins TW (2001) Investigating the neurocognitive deficits associated with chronic drug misuse. Curr Opin Neurobiol 11:250–257
Room P, Groenewegen HJ (1986) Connections of the parahippocampal cortex in the cat. II. Subcortical afferents. J Comp Neurol 251:451–473
Roozendaal B, Nguyen BT, Power AE, McGaugh JL (1999) Basolateral amygdala noradrenergic influence enables enhancement of memory consolidation induced by hippocampal glucocorticoid receptor activation. Proc Natl Acad Sci U S A 96:11642–11647
Sarnyai Z, Mello NK, Mendelson JH, Eros-Sarnyai M, Mercer G (1996) Effects of cocaine on pulsatile activity of hypothalamic–pituitary–adrenal axis in male rhesus monkeys: neuroendocrine and behavioral correlates. J Pharmacol Exp Ther 277:225–234
Sora I, Hall FS, Andrews AM, Itokawa M, Li XF, Wei HB, Wichems C, Lesch KP, Murphy DL, Uhl GR (2001) Molecular mechanisms of cocaine reward: combined dopamine and serotonin transporter knockouts eliminate cocaine place preference. Proc Natl Acad Sci U S A 98:5300–5305
Spealman RD (1995) Noradrenergic involvement in the discriminative stimulus effects of cocaine in squirrel monkeys. J Pharmacol Exp Ther 275:53–62
Tejani-Butt SM (1992) [3H]nisoxetine: a radioligand for quantitation of norepinephrine uptake sites by autoradiography or by homogenate binding. J Pharmacol Exp Ther 260:427–436
Teoh SK, Sarnyai Z, Mendelson JH, Mello NK, Springer SA, Sholar JW, Wapler M, Kuehnle JC, Gelles H (1994) Cocaine effects on pulsatile secretion of ACTH in men. J Pharmacol Exp Ther 270:1134–1138
Terenzi MG, Ingram CD (1995) A combined immunocytochemical and retrograde tracing study of noradrenergic connections between the caudal medulla and bed nuclei of the stria terminalis. Brain Res 672:289–297
van Groen T, Wyss JM (1990) The connections of presubiculum and parasubiculum in the rat. Brain Res 518:227–243
Volkow ND, Wang GJ, Fischman MW, Foltin RW, Fowler JS, Abumrad NN, Vitkun S, Logan J, Gatley SJ, Pappas N, Hitzemann R, Shea CE (1997) Relationship between subjective effects of cocaine and dopamine transporter occupancy. Nature 386:827–830
Yeh SY, De Souza EB (1991) Lack of neurochemical evidence for neurotoxic effects of repeated cocaine administration in rats on brain monoamine neurons. Drug Alcohol Depend 27:51–61
Zhu MY, Shamburger S, Li J, Ordway GA (2000) Regulation of the human norepinephrine transporter by cocaine and amphetamine. J Pharmacol Exp Ther 295:951–959
Acknowledgements
This study was supported by a grant (DA09085) from the National Institute on Drug Abuse.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beveridge, T.J.R., Smith, H.R., Nader, M.A. et al. Effects of chronic cocaine self-administration on norepinephrine transporters in the nonhuman primate brain. Psychopharmacology 180, 781–788 (2005). https://doi.org/10.1007/s00213-005-2162-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-2162-1