Abstract
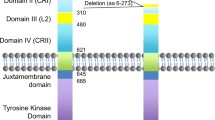
The naturally occurring mutated form of the epidermal growth factor receptor, ΔEGFR (also named EGFRvIII and de2-7EGFR), greatly enhances glioblastoma (GBM) cell growth in vivo through several activities, such as down-regulating p27 and up-regulating BclX(L) while increasing signaling through the RAS-MAPK and PI3-K cascades. More than half of GBMs, especially of the de novo type, overexpress EGFR, and 50%–70% of these express ΔEGFR. However, little is known about the distribution of ΔEGFR-expressing tumor cells within surgical specimens. In order to address this clinically important issue, we performed immunohistochemical analyses of 53 GBMs obtained during surgery using the anti-ΔEGFR monoclonal antibody, DH8.3. We also simultaneously analyzed wild-type EGFR expression in these tissues using the anti-EGFR monoclonal antibody, EGFR. 113. ΔEGFR and wild-type EGFR expression were observed in 20/53 (38%) and 29/53 (55%), respectively. Nineteen (95%) of the ΔEGFR-positive tumors also expressed wild-type EGFR; one case was ΔEGFR-positive but wild-type EGFR-negative. In 13/20 (65%) of the ΔEGFR-positive tumors, tumor cells were scattered diffusely within the tumors, 6/20 showed geographical distribution of ΔEGFR-positive tumor cells, and one case showed homogeneous staining. In the wild-type EGFR-positive cases, almost all tumor cells expressed EGFR. The differential distribution of cells expressing the two receptors observed here may suggest either that ΔEGFR arises at a low frequency from wild-type EGFR-expressing cells, perhaps during the process of gene amplification, or that there is a paracrine-type of interaction between them.
Similar content being viewed by others
References
Wong AJ, Bigner SH, Bigner DD, et al (1987) Increased expression of the epidermal growth factor receptor gene in malignant gliomas is invariably associated with gene amplification. Proc Natl Acad Sci USA 84:6899–6903
Ekstrand AJ, Sugawa N, James CD, et al (1992) Amplified and rearranged epidermal-growth-factor-receptor genes in human glioblastomas reveal deletions of sequences encoding portions of the N-and/or C-terminal tails. Proc Natl Acad Sci USA 89:4309–4313
Simmons ML, Lamborn KR, Takahashi M, et al (2001) Analysis of complex relationships between age, p53, epidermal growth factor receptor, and survival in glioblastoma patients. Cancer Res 61:1122–1128
Wikstrand CJ, Hale LP, Batra SK, et al (1995) Monoclonal antibodies against EGFRvIII are tumor specific and react with breast and lung carcinoma and malignant gliomas. Cancer Res 55:3140–3148
Feldkamp MM, Lala P, Lau N, et al (1999) Expression of activated epidermal growth factor receptors, Ras-guanosine triphosphate, and mitogen-activated protein kinase in human glioblastoma multiforme specimens. Neurosurgery 45:1441–1453
Frederick L, Wang X-Y, Eley G, et al (2000) Diversity and frequency of epidermal growth factor receptor mutations in human glioblastomas. Cancer Res 60:1383–1387
Nishikawa R, Ji XD, Harmon RC, et al (1994) A mutant epidermal growth factor receptor common in human glioma confers enhanced tumorigenicity. Proc Natl Acad Sci USA 91:7727–7731
Huang H-JS, Nagane M, Klingbeil CK, et al (1997) The enhanced tumorigenic activity of a mutant epidermal growth factor receptor common in human cancers is mediated by threshold levels of constitutive tyrosine phosphorylation and unattenuated signaling. J. Biol Chem 272:2027–2935
Wikstrand CJ, McLendon RE, Friedman AH, et al (1997) Cell surface localization and density of the tumor-associated variant of the epidermal growth factor receptor, EGFRvIII. Cancer Res 57: 4130–4140
Narita Y, Nagane M, Mishima K, et al (2002) Mutant epidermal growth factor receptor signaling down-regulates p27 through activation of the phosphatidylinositol 3-kinase/Akt pathway in glioblastomas. Cancer Res 62:6764–6769
Nagane M, Coufal F, Lin H, et al (1996) A common mutant epidermal growth factor receptor confers enhanced tumorigenicity on human glioblastoma cells by increasing proliferation and reducing apoptosis. Cancer Res 56:5079–5086
Lal A, Glazer CA, Martinson HM, et al (2002) Mutant epidermal growth factor receptor up-regulates molecular effectors of tumor invasion. Cancer Res 62:3335–3339
Kleihues P, Burger PC, Collins VP, et al (2000) Glioblastoma. In: Kleihues P, Cavenee WK (eds) Pathology and genetics of tumours of the nervous system. IARC, Lyon, pp 29–39
Hills D, Rowlinson-Busza G, Gullick WJ (1995) Specific targeting of a mutant, activated EGF receptor found in glioblastoma using a monoclonal antibody. Int J Cancer 63:537–543
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishikawa, R., Sugiyama, T., Narita, Y. et al. Immunohistochemical analysis of the mutant epidermal growth factor, ΔEGFR, in glioblastoma. Brain Tumor Pathol 21, 53–56 (2004). https://doi.org/10.1007/BF02484510
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02484510