Abstract
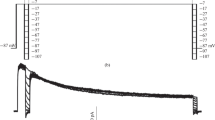
Inward rectifying potassium single-channel currents were studied in the membrane of guinea pig cardiac myocytes. In isolated inside-out patches two different channels were observed: a channel of 25 pS conductance ([K+]o=147 mM,T=21° C), if the solution at the cytoplasmic face of the patch contained 4 mM ATP and a channel of 80 pS conductance without ATP. The 25-pS-channel was also regularly seen in cell-attached patches (Sakmann and Trube 1984a, b), but the 80-pS-channel appeared only after inhibiting cellular metabolism by DNP. The percentage of time which the 25-pS-channel spent in the open state was 3.3 times larger in isolated patches compared to cell-attached patches. However, both types of single channel currents disappeared several minutes after the isolation of the patches.
In contrast to the 25-pS-channel, the 80-pS-channel, which is activated by the lack of ATP, carried measurable outward currents saturating at 1.5 pA (inward rectification). It is suggested that the 80-pS-channel mediates part of the increase in potassium current during metabolic inhibition. The openings of this channel appeared in bursts. The mean open time was 1.6 ms and the mean duration of the gaps within bursts 0.33 ms at −80 mV.
Article PDF
Similar content being viewed by others
References
Barrett JN, Magleby KL, Pallotta BS (1982) Properties of single calcium-activated potassium channels in cultured rat muscle. J Physiol 331:211–230
Berman KM, Cohn M (1970) Phosphoenolpyruvate synthetase: partial reactions studied with adenosine triphosphate analogues and the inorganic phosphate-H2 15O exchange reaction. J Biol Chem 245:5319–5325
Bers DM (1982) A simple method for the accurate determination of free [Ca] in Ca-EGTA solutions. Am J Physiol 242:C404-C408
Carmeliet E (1978) Cardiac transmembrane potentials and metabolism. Circ Res 42:577–587
Cavalié A, Ochi R, Pelzer D, Trautwein W (1983) Elementary currents through Ca2+ channels in guinea pig myocytes. Pflügers Arch 398:284–297
Colquhoun D, Neher E, Reuter H, Stevens CF (1981) Inward current channels activated by intracellular Ca in cultured cardiac cells. Nature 294:752–754
Ellis D, Thomas RC (1976) Direct measurement of the intracellular pH of mammalian cardiac muscle. J Physiol 262:755–771
Fink R, Hase S, Lüttgau HCh, Wettwer E (1983) The effect of cellular energy reserves and internal calcium ions on the potassium conductance in skeletal muscle of the frog. J Physiol 336:211–228
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391:85–100
Haas HG, Kern R, Einwächter HM (1970) Electrical activity and metabolism in cardiac tissue: an experimental and theoretical study. J Membr Biol 3:180–209
Hess P, Metzger P, Weingart R (1982) Free magnesium in sheep, ferret and frog striated muscle at rest measured with ion-selective micro-electrodes. J Physiol 333:173–188
Isenberg G (1977) Cardiac Purkinje fibres. [Ca2+] controls steady state potassium conductance. Pflügers Arch 371:71–76
Isenberg G, Klöckner U (1982) Calcium tolerant ventricular myocytes prepared by preincubation in a “KB-medium”. Pflügers Arch 395:6–18
Isenberg G, Vereecke J, van der Heyden G, Carmeliet E (1983) The shortening of the action potential by DNP in guinea pig ventricular myocytes in mediated by an increase of a time-independent K conductance. Pflügers Arch 397:251–259
Martell AE, Schwarzenbach G (1956) Adenosinphosphate und Triphosphat als Komplexbildner für Calcium und Magnesium. Helv Chim Acta 39:653–661
Matthews PM, Radda GK, Taylor DJ (1981) A31P n.m.r. study of metabolism in the hypoxic perfused rat heart. Trans Biochem Soc 9:236–237
McLeod DP, Daniel EE (1965) Influence of glucose on the transmembrane action potential of anoxic papillary muscle. J Gen Physiol 48:887–899
Methfessel C, Boheim G (1982) The gating of single calcium-dependent potassium channels is described by an activation/blockade mechanism. Biophys Struct Mech 9:35–60
Noma A (1983) ATP-regulated K+ channels in cardiac muscle. Nature 305:147–148
Piper HM, Probst I, Schwartz P, Hütter FJ, Spieckermann PG (1982) Culturing of calcium stable adult cardiac myocytes. J Mol Cell Cardiol 14:397–412
Sakmann B, Neher E (1983) Geometric parameters of pipettes and membrane patches. In: Sakmann B, Neher E (eds) Single channel recording. Plenum Press, New York, pp 37–51
Sakmann B, Trube G (1984a) Conductance properties of single inwardly rectifying potassium channels in ventricular cells from guinea pig heart. J Physiol 347:641–657
Sakmann B, Trube G (1984b) Voltage-dependent inactivation of inward-rectifying single-channel currents in the guinea pig heart cell membrane. J Physiol 347:659–683
Sheu SS, Fozzard HA (1982) Transmembrane Na+ and Ca2+ electrochemical gradients in cardiac muscle and their relationship to force development. J Gen Physiol 80:325–351
Trautmann A, Siegelbaum SA (1983) The influence of membrane patch isolation on single acetylcholine-channel current in rat myotubes. In: Sakmann B, Neher E (eds) Single channel recording. Plenum Press, New York, pp 473–480
Trube G, Hescheler J (1983) Potassium channels in isolated patches of cardiac cell membrane. Naunyn-Schmiedeberg's Arch Pharmacol 322:R64
Vleugels A, Vereecke J, Carmeliet E (1980) Ionic currents during hypoxia in voltage-clamp cat ventricular muscle. Circ Res 47:501–508
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trube, G., Hescheler, J. Inward-rectifying channels in isolated patches of the heart cell membrane: ATP-dependence and comparison with cell-attached patches. Pflugers Arch. 401, 178–184 (1984). https://doi.org/10.1007/BF00583879
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583879