Summary
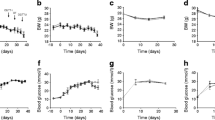
The role of glucagon in diabetic hyperglycaemia has been a matter of controversy because of difficulties in the production of selective glucagon deficiency. We developed a high-capacity (40 nmol/ ml), high-affinity (0.6·1011 l/mol) monoclonal glucagon antibody (Glu-mAb) and gave i.v. injections (4 ml/kg) to rats in order to study the effect of selective glucagon deficiency on blood glucose. Controls received a mAb against trinitrophenyl. Glu-mAb completely abolished the hyperglycaemic effect of 2.86 nmol/kg glucagon in normal rats (p<0.05, n=6). In moderately hyperglycaemic rats injected with streptozotocin as neonates (N-STZ), Glu-mAb abolished a postprandial increase in blood glucose (from 11.2±0.7 mmol/l to 17.3±1.8 mmol/l in controls vs 10.5±0.9 mmol/l to 9.3±1.0 mmol/l; cross-over: n=6, p<0.05). No significant effect of Glu-mAb treatment was observed in more hyperglycaemic N-STZ rats (cross-over, n=4) and in severely hyperglycaemic rats injected with STZ as adults (n=6), but after insulin treatment of the latter, at doses partially restoring blood glucose levels (12.7±4.3 mmol/l), Glu-mAb administration almost normalized blood glucose (maximal difference: 6.0±3.8 mmol/l; cross-over: n=5, p<0.05). In conclusion, our results provide strong additional evidence for the hypothesis that glucagon is involved in the pathogenesis of diabetes. The hormone plays an important role in the development of STZ-diabetic hyperglycaemia, but glucagon neutralization only leads to normoglycaemia in the presence of insulin.
Article PDF
Similar content being viewed by others
Abbreviations
- A-STZ rats:
-
Adult streptozotocin injected rats
- AUC:
-
area under the curve
- BW:
-
body weight
- Con-mAb:
-
monoclonal control antibody
- Glu-mAb:
-
monoclonal anti-glucagon antibody
- Ka :
-
constant of association
- NIDDM:
-
non-insulin-dependent diabetes mellitus
- N-STZ rats:
-
neonatal streptozotocin-injected rats
- OGTT:
-
oral glucose tolerance test
- STZ:
-
streptozotocin
References
DeFronzo RA, Bonadonna RC, Ferrannini E (1992) Pathogenesis of NIDDM. A balanced overview. Diabetes Care 15: 318–368
Unger RH, Orci L (1975) The essential role of glucagon in the pathogenesis of diabetes mellitus. Lancet I: 14–16
Dobbs RE, Sakurai H, Sasaki H et al. (1975) Glucagon: role in the hyperglycemia of diabetes mellitus. Science 187: 544–547
Gerich JE, Lorenzi M, Bier DM et al. (1975) Prevention of human diabetic ketoacidosis by somatostatin. Evidence for an essential role of glucagon. N Engl J Med 292: 985–989
Schusdziarra V, Rivier R, Dobbs RE, Brown M, Vale W, Unger RH (1978) Somatostatin analogs as glucagon suppressants in diabetes. Horm Metab Res 10: 563–565
Santeusanio F, Massi-Benedetti M, Angeletti G, Calabrese G, Bueti A, Brunetti P (1981) Glucagon and carbohydrate disorder in totally pancreatectomized man (A study with the aid of an artificial endocrine pancreas). J Endocrinol Invest 4: 93–96
Flatt PR, Swanston-Flatt SK, Bailey CJ (1979) Glucagon antiserum: a tool to investigate the role of circulating glucagon in obese-hyperglycaemic (ob/ob) mice. Biochem Soc Trans 7: 911–913 (Abstract)
Unson CG, Gurzenda EM, Merrifield RB (1989) Biological activities of des-His1[Glu9] glucagon amide, a glucagon antagonist. Peptides 10: 1171–1177
Almdal TP, Holst JJ, Heindorff H, Vilstrup H (1992) Glucagon immunoneutralization in diabetic rats normalizes urea synthesis and decreases nitrogen wasting. Diabetes 41: 12–16
Unger RH (1978) Role of glucagon in the pathogenesis of diabetes: the status of the controversy. Metabolism 27: 1691–1709
Sherwin RS, Felig P (1978) Hyperglucagonemia in diabetes. N Engl J Med 299: 1366–1368 (Letter)
Müller WA (1978) Diabetes mellitus, eine bihormonale Krankheit? Dtsch Med Wschr 103: 1219–1221
Buchanan KD (1977) Glucagon. In: Bajaj JS (ed) Insulin and metabolism. Excerpta Medica, London, pp 233–270
Barnes AJ, Bloom SR, Alberti KGMM, Smythe P, Alford FP, Chisholm DJ (1977) Ketoacidosis in pancreatectomized man. N Engl J Med 296: 1250–1253
Barnes AJ, Kohner EM, Bloom SR, Johnston DG, Alberti KGMM, Smythe P (1978) Importance of pituitary hormones in aetiology of diabetic ketoacidosis. Lancet I: 1171–1174
Consoli A, Nurjhan N, Reilly JJ, Bier DM, Gerich JE (1990) Mechanism of increased gluconeogenesis in noninsulin-dependent diabetes mellitus. J Clin Invest 86: 2038–2045
Reaven GM, Chen Y-DI, Golay A, Swislocki ALM, Jaspan JB (1987) Documentation of hyperglucagonemia throughout the day in nonobese and obese patients with noninsulin-dependent diabetes mellitus. J Clin Endocrinol Metab 64: 106–110
Consoli A, Nurjhan N, Capani F, Gerich JE (1989) Predominant role of gluconeogenesis in increased hepatic glucose production in NIDDM. Diabetes 38: 550–557
Baron AD, Schaeffer L, Shragg P, Kolterman OG (1987) Role of hyperglucagonemia in maintenance of increased rates of hepatic glucose output in type II diabetes. Diabetes 36: 274–283
DeFronzo RA, Ferrannini E, Simonson DC (1989) Fasting hyperglycemia in non-insulin-dependent diabetes mellitus: contributions of excessive hepatic glucose production and impaired tissue glucose-uptake. Metabolism 4: 387–395
Campbell DG, Mandarino LJ, Gerich JE (1988) Quantification of the relative impairment in actions of insulin on hepatic glucose production and peripheral glucose uptake in noninsulin-dependent diabetes mellitus. Metabolism 37: 15–22
Alberti KGMM, Christensen NJ, Iversen J, Ørskov H (1975) Role of glucagon and other hormones in the development of diabetic ketoacidosis. Lancet I: 1307–1311
Barnes AJ, Crowley MF, Bloom A et al. (1975) Is glucagon important in stable insulin-dependent diabetes. Lancet II: 734–737
Gerich JE (1981) Somatostatin: In: Brownlee M (ed) Handbook of diabetes mellitus. Vol. 1. Garland STPM Press, New York, pp 297–354
Goldberg DJ, Walesky M, Sherwin RS (1979) Effects of somatostatin on the plasma amino acid response to ingested protein in man. Metabolism 28: 866–872
Meneilly GS, Minaker KL, Elahi D, Rowe JW (1988) Somatostatin infusion enhances hepatic glucose production during hyperglucagonemia. Metabolism 37: 252–256
Valverde I, Alarcon C, Ruiz-Grande C, Rovira A (1988) Plasma glucagon and glucagon-like immunoreactivity in totally pancreatectomized humans. In: Tiengo A, Alberti KGMM, Del Prato S, Vranic M (eds) Diabetes secondary to pancreatopathy. Excerpta Medica, Amsterdam New York Oxford, pp 51–62
Holst JJ, Galbo H, Richter EA (1978) Neutralization of glucagon by antiserum as a tool in glucagon physiology. J Clin Invest 62: 182–190
Thim L, Moody AL (1981) The primary structure of porcine glicentin (proglucagon). Regul Pept 2: 139–151
Köhler G, Milstein C (1976) Derivation of specific antibody-producing tissue culture and tumor cell lines by cell fusion. Eur J Immunol 6: 511–519
Holst JJ, Aasted B (1974) Production and evaluation of glucagon antibodies for radioimmunoassay. Acta Endocrinol 77: 715–726
Rasch R (1979) Control of blood glucose levels in the streptozotocin diabetic rat using a long-acting heat-treated insulin. Diabetologia 16: 185–190
Lisato G, Cusin I, Tiengo A, Del Prato S, Jeanrenaud B (1992) The contribution of hyperglycaemia and hypoinsulinaemia to insulin resistance of streptozotocin-diabetic rats. Diabetologia 35: 310–315
Rodbell M, Krans HMJ, Pohl SL, Birnbaumer L (1971) The glucagon-sensitive adenyl cyclase system in plasma membranes of rat liver. J Biol Chem 246: 1861–1871
Jelinek LJ, Lok S, Rosenberg GB et al. (1993) Expression cloning and signaling properties of the rat glucagon receptor. Science 259: 1614–1616
Barling P, Beloff-Chain A (1973) Studies on the administration of glucagon and insulin antibodies to rats. Horm Metab Res 5: 154–159
Heindorff H, Holst JJ, Almdal T, Vilstrup H (1993) Effect of glucagon immunoneutralization on the increase in urea synthesis after hysterectomy in rats. Eur J Clin Invest 23: 166–171
Luyckx AS (1974) Etude de la sécrétion de l'insulin et du glucagon. Faculté de Médicine, Université de Liege, Liege pp 122
Frohman LA, Reichlin M, Sokal JE (1970) Immunologic and biologic properties of antibodies to a glucagon-serum albumin polymer. Endocrinology 87: 1055–1061
Tan K, Tsiolakis D, Marks V (1985) Effect of glucagon antibodies on plasma glucose, insulin and somatostatin in the fasting and fed rat. Diabetologia 28: 435–440
Malaisse WJ, Giroix M-H, ZÄhner D, Marynissen G, Sener A, Portha B (1991) Neonatal streptozotocin injection: a model of glucotoxicity. Metabolism 40: 1101–1105
Lickley HLA, Kemmer FW, Doi K, Vranic M (1983) Glucagon suppression improves glucoregulation in moderate but not chronic severe diabetes. Am J Physiol 245: E424-E429
Doi K, Prentki M, Yip C, Müller WA, Jeanrenaud B, Vranic M (1979) Identical biological effects of pancreatic glucagon and a purified moiety of canine gastric immunoreactive glucagon. J Clin Invest 63: 525–531
McGarry JD, Foster DW (1983) Glucagon and ketogenesis. In: Lefèbvre PJ (ed) Handbook of experimental pharmacology, Glucagon 66/I. Springer, Berlin Heidelberg New York, pp 383–398
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brand, C.L., Rolin, B., JØrgensen, P.N. et al. Immunoneutralization of endogenous glucagon with monoclonal glucagon antibody normalizes hyperglycaemia in moderately streptozotocin-diabetic rats. Diabetologia 37, 985–993 (1994). https://doi.org/10.1007/BF00400461
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400461