Summary
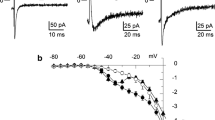
A fast perfusion system was used to analyze the kinetics of the response of L-type calcium current (ICa) to rapid exposures to β-adrenergic or dihydropyridine agonists in whole-cell patch-clamped frog ventricular myocytes. The perfusion system was based on the lateral motion of an array of plastic capillary tubes from which solutions flowed at a velocity of ∼ 5 cm/s. Movement from one capillary to the adjacent one occurred in < 20 ms and complete exchange of extracellular solution was achieved in < 50 ms as demonstrated by the block of ICa by fastflow application of Cd during a depolarizing pulse. Fastflow applications of increasing concentrations of isoprenaline (Iso) led to a dose-dependent stimulation of ICa at [Iso] > 1 nM. The response of ICa to Iso always started after a delay of several seconds. The delay duration decreased as [Iso] increased, and was typically ∼ 3 s at 10 μM Iso. The rising phase of ICa increase was monophasic and independent of [Iso] > 100 nM. For short applications of Iso (8.8 s), half maximal and maximal stimulation of ICa occurred ∼ 20 s and ∼ 40 s after the beginning of Iso application, respectively. When Iso was applied during a depolarizing pulse (with Ba as the charge carrier), IBa never increased during that pulse. The kinetics of the ICa response to Iso were not affected by varying the voltage clamp protocols or the ionic composition of intracellular and extracellular solutions. In comparison with the effects of Iso, the stimulatory effect of the dihydropyridine agonist (−)Bay K 8644 on ICa was ∼ 15 times faster: delay, half-time to maximal and time to maximal responses were 15 times shorter with (−)Bay K 8644 than with Iso. It is concluded that frog ventricular myocytes respond slowly to a quick application of β-adrenergic agonists.
Similar content being viewed by others
References
Alloatti G, Serazzi L, Levi RC (1991) Prostaglandin-I2 (PGI2) enhances calcium current in guinea-pig ventricular heart cells. J Mol Cell Cardiol 23:851–860
Argibay JA, Fischmeister R, Hartzell HC (1988) Inactivation, reactivation and pacing dependence of calcium current in frog cardiocytes: correlation with current density. J Physiol 401:201–226
Boutjdir M, Méry P-F, Hanf R, Shrier A, Fischmeister R (1990) High affinity forskolin inhibition of L-type Ca2+ current in cardiac cells. Mol Pharmacol 38:758–765
Breitweiser GE (1991) G-protein mediated ion channel activation. Hypertension 17:684–692
Breitweiser GE, Szabo G (1985) Uncoupling of cardiac muscarinic and β-adrenergic receptors from ion channels by a guanine nucleotide analogue. Nature 317:538–540
Carbone E, Swandulla D (1989) Neuronal calcium channels: kinetics, blockade, and modulation. Prog Biophys Mol Biol 54:31–58
Cavalié A, Allen TJ, Trautwein W (1991) Role of the GTP-binding protein Gs in the β-adrenergic modulation of cardiac Ca channels. Pflügers Arch 419:433–444
Dolphin AC (1991) Regulation of calcium channel activity by GTP binding proteins and second messengers. Biochim Biophys Acta 1091:68–80
Dunlap K, Holz GG, Rane SG (1987) G proteins as regulators of ion channel function. Trends Neurosci 10:241–244
Fischmeister R, Hartzell HC (1986) Mechanism of action of acetylcholine on calcium current in single cells from frog ventricle. J Physiol 376:183–202
Fischmeister R, Hartzell HC (1987) Cyclic guanosine 3′,5′-monophosphate regulates the calcium current in single cells from frog ventricle. J Physiol 387:453–472
Fischmeister R, Shrier A (1989) Interactive effects of isoprenaline, forskolin and acetylcholine on Ca2+ current in single frog ventricular cells. J Physiol 417:213–239
Gilman AG (1987) G proteins: transducers of receptor-generated signals. Ann Rev Biochemistry 56:615–649
Godt RE, Lindley BD (1982) Influence of temperature upon contractile activation and isometric force production in mechanically skinned muscle fibers of the frog. J Gen Physiol 80:279–297
Hartzell HC (1988) Regulation of cardiac ion channels by catecholamines, acetylcholine and 2nd messenger systems. Prog Biophys Mol Biol 52:165–247
Hartzell HC, Fischmeister R (1986) Opposite effects of cyclic GMP and cyclic AMP on Ca2+ current in single heart cells. Nature 323:273–275
Hartzell HC, Fischmeister R (1992). Direct regulation of cardiac Ca channels by G proteins: neither proven nor needed? Trends Pharmacol Sci 13:380–385
Hartzell HC, Méry P-F, Fischmeister R, Szabo G (1991) Sympathetic regulation of cardiac calcium current is due exclusively to cAMP-dependent phosphorylation. Nature 351:573–576
Hescheler J, Kameyama M, Trautwein W, Mieskes G, Söling H-D (1987a) Regulation of the cardiac calcium channel by protein phosphatases. Eur J Biochem 165:261–266
Hescheler J, Tang M, Jastorff B, Trautwein W (1987b) On the mechanism of histamine induced enhancement of the cardiac Ca2+ current. Pflügers Arch 410:23–29
Holmer SR, Homey CJ (1991) G proteins in the heart. A redundant and diverse transmembrane signaling network. Circulation 84: 1891–1902
Holz GG, Rane SG, Dunlap K (1986) GTP-binding proteins mediate transmitter inhibition of voltage-dependent calcium channels. Nature 319:670–672
Imoto Y, Yatani A, Reeves JP, Codina J, Birnbaumer L, Brown AM (1988) α-subunit of Gs directly activates cardiac calcium channels in lipid bilayers. Am J Physiol 255:H722-H728
Kameyama M, Hofmann F, Trautwein W (1985) On the mechanism of β-adrenergic regulation of the Ca channel in the guinea-pig heart. Pflügers Arch 405:285–293
Kameyama M, Hescheler J, Hofmann F, Trautwein W (1986a) Modulation of Ca current during the phosphorylation cycle in the guinea pig heart. Pflügers Arch 407:123–128
Kameyama M, Hescheler J, Mieskes MG, Trautwein W (1986b) The protein-specific phosphatase 1 antagonizes the β-adrenergic increase of the cardiac Ca current. Pflügers Arch 407:461–463
Kozlowski RZ, Twist VW, Brown AM, Powell T (1991) Flash photolysis of intracellular caged GTPγS increases L-type Ca2+ currents in cardiac myocytes. Am J Physiol 261:H1665-H1670
Kurachi Y, Tung RT, Ito H, Nakajima T (1992) G protein activation of cardiac muscarinic K+ channels. Prog Neurobiol 39:229–246
Levi RC, Alloatti G (1988) Histamine modulates calcium current in guinea-pig ventricular myocytes. J Pharmacol Exp Ther 246:377–383
Mattera R, Graziano MP, Yatani A, Zhou ZM, Graf R, Codina J, Birnbaumer L, Gilman AG, Brown AM (1989) Splice variants of the α-subunit of the G protein Gs activate both adenylyl cyclase and calcium channels. Science 243:804–807
Méry P-F, Brechler V, Pavoine C, Pecker F, Fischmeister R (1990) Glucagon stimulates the cardiac Ca2+ current by activation of adenylyl cyclase and inhibition of phosphodiesterase. Nature 345:158–161
Méry P-F, Lechêne P, Fischmeister R (1992) A loudspeaker-driven system for rapid and multiple solution exchanges in patch-clamp experiments. Pflügers Arch 420:529–535
Nakajima T, Wu S, Irisawa H, Giles W (1990) Mechanism of acetylcholine-induced inhibition of Ca current in bullfrog atrial myocytes. J Gen Physiol 96:865–885
O'Rourke B, Backx PH, Marban E (1992) Phosphorylation-independent modulation of L-type calcium channels by magnesium-nucleotide complexes. Science 257:245–248
Parsons TD, Lagrutta A, White RE, Hartzell HC (1991) Regulation of Ca2+ current in frog ventricular cardiomyocytes by 5′-guanylyl-imidodiphosphate and acetylcholine. J Physiol 432:593–620
Pelzer S, Shuba YM, Asai T, Codina J, Birnbaumer L, McDonald TF, Pelzer D (1990) Membrane-delimited stimulation of heart cell calcium current by β-adrenergic signal-transducing Gs protein. Am J Physiol 259:H264-H267
Pfaffinger PJ, Martin JM, Hunter DD, Nathanson NM, Hille B (1985) GTP-binding proteins couple cardiac muscarinic receptors to a K channel. Nature 317:536–538
Pfeuffer T, Helmreich EJM (1988) Structural and functional relationships of guanosine triphosphate binding proteins. Curr Top Cell Reg 29:129–216
Rodbell M (1980) The role of hormone receptors and GTP-regulatory proteins in membrane transduction. Nature 284:17–22
Shuba YM, Hesslinger B, Trautwein W, McDonald TF, Pelzer D (1990) Whole-Cell calcium current in guinea-pig ventricular myocytes dialysed with guanine nucleotides. J Physiol 424:205–228
Sternweis PC, Pang I-H (1990) The G protein-channel connection Trends Neurosci 13:122–126
Szabo G, Otero AS (1990) G-protein mediated regulation of K+ channels in heart. Ann Rev Physiol 52:293–305
Trautwein W, Hescheler J (1990) Regulation of cardiac L-type calcium current by phosphorylation and G-proteins. Ann Rev Physiol 52:257–274
Tsien RW, Bean BP, Hess P, Lansman JB, Nilius B, Nowycky MC (1986) Mechanisms of calcium channel modulation by β-adrenergic agents and dihydropyridine calcium agonists. J Mol Cell Cardiol 18:691–710
Yatani A, Brown AM (1989) Rapid β-adrenergic modulation of cardiac calcium channel currents by a fast G protein pathway. Science 245:71–74
Yatani A, Codina J, Imoto Y, Reeves JP, Birnbaumer L, Brown AM (1987) A G protein directly regulates mammalian cardiac calcium channels. Science 238:1288–1291
Yatani A, Imoto Y, Codina J, Hamilton SL, Brown AM, Birnbaumer L (1988) The stimulatory G protein of adenylyl cyclase, Gs, also stimulates dihydropyridine-sensitive Ca2+ channels. Evidence for direct regulation independent of phosphorylation by cAMP-dependent protein kinase or stimulation by a dihydropyridine agonist. J Biol Chem 263:9887–9895
Author information
Authors and Affiliations
Additional information
Correspondence to: R. Fischmeister at the above address
Rights and permissions
About this article
Cite this article
Méry, PF., Frace, A.M., Hartzell, H.C. et al. A comparative analysis of the time course of cardiac Ca2+ current response to rapid applications of β-adrenergic and dihydropyridine agonists. Naunyn-Schmiedeberg's Arch Pharmacol 348, 197–206 (1993). https://doi.org/10.1007/BF00164799
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00164799